Are We at the Very Exhilarating Moment of Moving from Treating to Curing Autoimmune Diseases? // Important Ethical/Clinical Issues for Fertility Practice
Today’s postings address two main subject, first reproductive immunology – where amazing things are happening - with special reference to autoimmune diseases. And, secondly, we are re-presenting two issues of considerable clinical as well as medico-ethical relevance, - what should the limits be for gamete donors (eggs or semen) in how many pregnancies they should be allowed to contribute to and – the second issue - the commercial offering by a small number of laboratories and IVF clinics the so-called polygenic risk scoring of embryos (also called PGT-P), which an ASRM opinion just rightly exposed as something that can only be described as a “snake oil offering.” As always, the CHR does not mince words and – this time – we are especially pleased to speak out because it is a pleasure to have the ASRM seemingly on our side.
The CHR’s Editorial Staff
Are We at the Very Exhilarating Moment of Moving from Treating to Curing Autoimmune Diseases?
The Editorial Staff of the CHR
One of today’s main topic is immunology, and especially autoimmunity. Eric Topol in one of his recent Substack postings, GroundTruth, noted that the treatment of autoimmune diseases is at an “exhilarating” turning point, switching away from nonspecific immune system suppression to elimination of disease causes. He in that posting then also reminded us that autoimmune diseases have doubled in prevalence after the COVID-19 pandemic, - implying that curing autoimmune diseases is more urgent than ever. Autoimmune diseases, of course, occupy an unusually close association with reproduction because both are dependent on a normally functioning immune system, able to induce tolerance. In pregnancy this means that the mother’s immune system must develop adequate tolerance toward the paternal allograft (the placental-fetal unit). And – if that does not happen, – we experience pregnancy losses and, possibly, also implantation failure. In autoimmunity a person’s inherent tolerance against “self” has broken down, resulting in that person’s immune system attacking a component of the patient’s own body. This is the reason why autoimmunity is such an important subject in reproductive medicine and why it is so difficult to understand that so many REI - colleagues still consider the maternal immune system to be largely irrelevant for fertility treatments. Here is, therefore, a short update on important new developments in autoimmunity with relevance for reproduction.
Here covered materials were already addressed in the January 2026 issue of the CHRVOICE but have been updated and reedited.
New Revolutionary Treatments for Autoimmune Diseases
Two recent developments lead Topol to the believe that cures are just around the corner: The first, called “hard reset,” is a new development initially reported by German researcher just approximately four years ago, in which they “stole” a brilliant idea from our oncology colleagues, - the use of engineered T cells, known as chimeric antigen receptor (CAR) cells. This treatment is described as hard reset because it depletes B cells and - once after depletion of “bad” B cell repopulated – they no longer are the self-attacking disease-causing immune cells they were before.
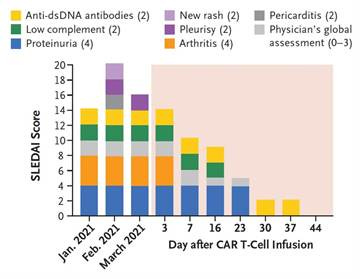
In the initial German experiment that started the hard reset treatments of autoimmunity, a 20-year-old woman with treatment -resistant systemic lupus erythematosus (SLE) received one single infusion of CAR T cells that targeted CD-19 on lymphocytes, and this cured the patients (at least so-far). The patient’s response to the treatment over only six weeks is depicted in the Figure below.
The German investigators have since achieved similar success with systemic sclerosis and idiopathic inflammatory myositis. As other investigators have started to apply this treatment – for several years before that in wide use in oncology especially in so-called liquid cancers (lymphomas, leukemias) - CAR T cell treatments now also is becoming a mainstay in autoimmunity, for the first time making it apparently possible to cure autoimmune diseases.
By now being already able to use allogeneic cells from healthy donors (in early stages of these treatments only autologous cells were used which had to be manufactured at very high cost outside the body) or from a master cell banks, everything now can be accomplished inside the patient’s own the body. Because this in vivo treatment no longer requires lymphocyte depletion (via chemotherapy) the treatment has become much less toxic and can be given on an outpatient basis. Several companies are, indeed, using this new approach, already in in vivo clinical trials for several autoimmune diseases.
In contrast to the hard reset, the so-called “soft reset,” follows a different approach in treating autoimmune diseases, producing inverse vaccines to induce tolerance. This second approach appears to us of special interest for reproductive immunologists and REIs because insufficient induction of anti-paternal tolerance is, of course, a frequent cause of repeated pregnancy loss. And the CHR wouldn’t be surprised if poor tolerance induction also played a role is some cases of so-called “unexplained infertility.”
The goal of a so-called tolerogenic vaccines is exactly the opposite of a traditional, standard vaccines, which have the purpose of strengthening the immune response against a given virus. Tolerogenic vaccines, of course, have the goal of weakening the immune response against “self-epitopes.” Autoimmune diseases already in clinical trials with such tolerogenic vaccines - according to Topol - are celiac disease, multiple sclerosis, and type 1 diabetes as well as rheumatoid arthritis.
Celiac Disease, - the Great Imitator
It is no coincidence that autoimmunity is such an important and frequently featured subject by the CHR because there is likely no other group of diseases which has as close an association with reproduction as autoimmune diseases. The reason is obvious and has been noted in these pages over and over again: The core issue in autoimmunity and pregnancy is the same: It is called immune -tolerance, - in pregnancy tolerance by the mother’s immune system of the fetal-placental unit (in most cases a paternal semi-allograft but increasingly frequently also a full allograft if donor eggs are used of if a gestational carrier carries the pregnancy) and in autoimmunity it is self-tolerance. If tolerance fails in pregnancy, the pregnancy fails. If self-tolerance fails, autoimmunity is the result.

There is so much more to say about this commonality between autoimmunity and pregnancy, first recognized by Professor James S. Scott (1924-2006) in the UK already in the 1960s and 1970s, in his obituary in the Independent correctly described as “having done as much as any obstetrician of his generation to unravel the diseases caused by defects in the immunological relationship between mother and baby.”2
The CHR’s Medical Director and Chief Scientist, Norbert Gleicher, MD, knew Scott quite well and considered him to be a prophetic mentor on this subject. He always tells the story that he witnessed Scott preaching to rheumatologists to join him in using the pregnancy model to learn to understand SLE (imagine how much Scott would have enjoyed the above discussed Topol Substack!)
A brief Medscape article by Kevin Fernando, MBChB, is meant to remind us that the autoimmune disease, celiac disease (CD), is frequently misdiagnosed and often outright missed as a diagnosis. This is highly relevant to any infertile patient population because CD is not rare. It affects approximately 1.4% of the population, and like most autoimmune diseases shows strong female preponderance (and these numbers very likely significant underestimate the real prevalence). Average age of (often late) diagnosis is 40s through 60s, therefore potentially defining most of the CHR’s patients in diagnostic ages.
CD also shows the additional characteristic of often occurring in combination with other autoimmune diseases. Its, likely, however, most tricky characteristic is its greatly variable symptomatology which makes the differential diagnosis with several other gastro-intestinal conditions difficult. Those can include irritable bowel syndrome (IBS) or even inflammatory bowel diseases. And – considering how frequently we diagnose CD for the first time in the CHR’s patient population - granted a much older population than at other IVF clinics,- it in quite a number of infertile women seems to be an often overlooked diagnosis.
REFERENCES
Topol E. Substak-GroundTruth; December 14, 2025. https://erictopol.substack.com/p/the-exhilirating-movement-from-treatment
Independent. October 2, 2006. Orbituary.
https://www.the-independent.com/news/obituaries/professor-james-scott-418521.html Accesses December 25, 2025.
Fernando K. Medscape.. December 5, 2025. https://www.medscape.com/viewarticle/1003090
Important Ethical/Clinical Issues for Fertility Practice
The Editorial Staff of the CHR
We are here discussing two important clinical issues with significant ethical relevance, - how many offspring a gamete donor (whether egg or semen donor) should be allowed to have and - second – the concept of embryo biopsy for polygenic risk determination (PGT-P). Both subjects have remained controversial, and we feel that publicizing the CHR’s positions on these two issues is of importance
Here presented articles were already published in the January 2026 issue of the CHRVOICE but have been updated and reedited.
To How Many Offspring Should a Gamete-donor – Whether Egg- or Sperm-donor – be allowed to contribute to?
That is a question that has “plagued” the infertility field since the inception of gamete donations. And guidelines from professional societies have not made the decisions easier because their cut-offs usually made little sense. Here are some examples: The ASRM suggests under current guidelines published in 2024:1
Institutions, clinics, and sperm banks should maintain sufficient records to allow a limit to be set for the number of pregnancies for which a given donor is responsible. It is difficult to provide a precise number of times that a given donor can be used because one must take into consideration the population base from which the donor is selected and the geographic area that may be served by a given donor. It has been suggested that in a population of 800,000, limiting a single donor to no more than 25 births would avoid any significant increased risk of inadvertent consanguineous conception. This suggestion may require modification when the population represents an isolated subgroup or when the specimens are distributed over a wide geographic area.
OOCYTE DONORS -- should be limited to six (6) treatment cycles per donor representing a total of ca. 120 eggs and at most 25 births.
The basis for this recommendation is rooted in concern over the cumulative risk for the donor after undergoing more than six ovarian stimulations and oocyte retrieval procedures. When splitting donor embryo batches, the potential risk of siblings in close geographic proximity should be considered.
SEMEN DONATIIONS – Interestingly, there are no specific recommendations for semen donors.
ESHRE in a Position Paper in 2025 published very different guidelines which under the title, “International Limits on the Number of Offspring Per Gamete Donor,”2. are still under membership review and only considered a draft. ESHRE in this document also recognized that gamete donation these days can often cross borders.
The debate in Europe was recently boosted by the discovery that a very active Europe-wide sperm donor through a European sperm bank (he fathered at least 197 children) turned out to be an unrecognized carrier for a cancer gene (Li - Fraumeni syndrome which carried a 90% lifetime risk of developing various cancers, including several childhood cancers) he passed on to offsprings conceived with his donor sperm,- several of whom developed cancers.3
In its pending policy paper ESHRE proposes an international limit on the number of offspring per donor. In contrast to ASRM guidelines, ESHR also proposes that limits be set in terms of families rather than individual children and proposes now a generous limit of 50 families but, ultimately, supports – after a run-in period – a limit of 15 families. ESHRE, moreover, proposes that gamete banks self-impose those limits immediately until governments and/or EU can legislate the process.
The European document also included a table that listed current maximal limits for gamete donors in many European countries. For oocyte donors the numbers were in countries that allow egg donation between 3 and 10, - and between 3 and 15 for sperm donors.
In short, - gamete third-party donation is in many ways still the Wild West and would – arguably – greatly benefit from a rational worldwide set of rules. That such rules are overdue is also demonstrated by a related phenomenon, - characterized by the sudden desire of some super-wealthy individuals to have huge numbers of children. Recent reports in the lay press, for example, reported on several Chinese billionaires with allegedly each claiming over 100 children, most delivered by surrogate or gestational carriers, while a recent report in The Wall Street Journal reported on Pavel Durov, the Russian founder of Telegram, who now resides in Dubai, and claims to have donated his semen to over 100 births. The girlfriend of one Chinese Billionaire, Xu Bo, alleged that he has worldwide over 300 children (a story we reported before) He claimed that he, one day, wanted his over 20 U.S. children to take over his business.
In comparison, Elon Musk, with his 14 children, has still lots of catch-up to do!
REFERENCES
Practice Committees of ASRM and SART. Fertil Steril 2024;122(5):799-813
ESHRE Draft document. https://www.eshre.eu/Europe/Position-statements/Stakeholder-review
Gallagher J, Truswll N. BBC. December 10, 2025. https://www.bbc.com/news/articles/ckgmy90z991o
We Welcome the Long Overdue Publication of an ASRM Ethics and Practice Committee Report on Polygenic Risk Screening
The CHR in late 2025 published in its two publications a “CHR Opinion” wondering what had happened to a many months earlier among members of ASRM circulating draft of an ASRM & SART policy statement regarding the practice of polygenic risk screening of embryos (PGT-P) in IVF cycles. With this document now finally published in December of 2025, - under only signature of the ASRM Ethics and Practice Committees (why SART dropped out was not announced), we found it important to reprint the ASRM’s formal summary opinion, as originally distributed by the ASRM in a press release.4 The full length document was, as always, published in Fertility & Sterility.5
PRESS RELEASE
December 8, 2025
Washington, DC—Fertility and Sterility, the flagship publication of the American Society for Reproductive Medicine (ASRM), has published a joint report from the Ethics and Practice Committees evaluating the use of preimplantation genetic testing for polygenic disorders, known as PGT-P. The report concludes that this emerging technology is not ready for clinical practice and should not be offered as a reproductive service at this time, given the predictive uncertainties of the results and the substantial ethical considerations that it raises.
PGT-P aims to estimate an embryo’s potential risk for developing certain multifactorial diseases such as hypertension, heart disease and diabetes, but current predictive models are limited by insufficient data, incomplete understanding of gene and environment interactions, and a lack of diverse genomic representation. These gaps mean that the scores produced may not translate into meaningful clinical guidance for patients or providers. The Committees also note that whether individuals will ultimately develop the conditions for which PGT-P is being offered can be significantly impacted by factors such as diet, lifestyle and future medical treatments, with polygenic risk scoring accounting for only a small percentage of clinical variation.
Key Findings:
· PGT-P is a nascent and unproven technology that should not be used clinically at this time.
· Current evidence does not support the predictive accuracy, safety, or clinical value of polygenic embryo screening.
· The technology risks misleading patients by overstating what polygenic risk scores can reliably determine.
· PGT-P raises significant ethical issues, including concerns about equity, autonomy, disease prioritization, and the potential for biased or incomplete risk assessments.
· Any use of PGT-P should occur only in research settings under Institutional Review Board oversight until scientific, clinical, ethical, and societal concerns are addressed.
·
Together, this analysis provides an ethics-based framework to guide clinicians, policymakers, patients and the public at large as they navigate the emergence of polygenic embryo screening. By outlining the current limitations and risks of PGT-P, the Ethics Committee and Practice Committee underscore the critical importance of rigorous research, transparent communication, and careful scientific review before this technology is considered for clinical use.
AND HOW THE CHR SEES IT -- The CHR fully agrees with the summary of the ASRM guideline as published. The ASRM is, indeed, to be congratulated on publishing this document which – we are convinced – faced strong opposition from the genetic testing industry which has started offering PGT-P already through several laboratories and IVF clinics. To publish this opinion was morally and ethically the correct decision.
It must be said loud and clear: It is bad enough that daily thousands of normal embryos are either not transferred and/or discarded for no good reason because of preimplantation genetic testing for aneuploidy (PGT-A), thereby adversely affecting cumulative pregnancy and live birth chances for thousands of infertile couples seeking out fertility treatments. It is simply incredible that the genetic testing industry (i.e., PGT laboratories and selected IVF clinics) now further enlarge the pool of unused embryos in IVF with still good pregnancy chances that are not used or disposed by offering to their patients PGT-P in addition to PGT-A. One can only hope that, for all the reasons so well summarized in the ASRM document here re-presented, these offerings will now, based on this ASRM publication, stop.
But ASRM opinions have not much mattered when it comes to PGT-A and we, therefore, are doubtful that this recent document will make much of a difference. The only opinion the genetic testing industry apparently does take seriously is that of plaintiff lawyers. Too bad!
REFERENCES
Practice Committees of ASRM and SART. Fertil Steril 2024;122(5):799-813
ESHRE Draft document. https://www.eshre.eu/Europe/Position-statements/Stakeholder-review
Gallagher J, Truswll N. BBC. December 10, 2025. https://www.bbc.com/news/articles/ckgmy90z991o
ASRM. Press relase. December 8, 2025. https://www.asrm.org/news-and-events/asrm-news/press-releasesbulletins/asrm-ethics-and-practice-committees-release-new-report-concluding-polygenic-embryo-screening-is-not-ready-for-clinical-use/
Ethics and Practice Committees of the ASRM. Fertil Steril 2025; 10.1016/j.fertnstert.2025.10.023; ahead of print.