INTERESTING RECENT NEWS REGARDING CHROMOSOMAL TESTING OF EMBRYOS IN IVF
It is certainly no longer a secret that we, here at the CHR, are not very friendly imposed toward preimplantation genetic testing for aneuploidy (PGT-A) and we believe for good reasons: The test in general does not improve IVF cycle outcomes and, indeed, in certain sub-groups of IVF patients actually reduces their pregnancy chances and adds significant cost to an already excruciatingly expensive IVF cycle.
The CHR has held this position since 2006 (hard-to-believe, we are celebrating our 20th anniversary this year!) after reanalyzing two early prospectively randomized studies of Belgian colleagues of what - then – was called preimplantation genetic screening (PGS). Both studies had not found promised IVF cycle outcome improvement PGS-proponents had widely touted and the CHR’s reanalysis of their data actually suggested that PGS may reduce IVF pregnancy chances in older women.
We, of course, were not the only ones who – ever since – against significant resistance from the “PGT-A industry,” had to prove that by the PGT-A industry promised improved IVF cycle outcomes really never materialized. We in the process defined PGT-A as a “test (or procedure) in search of an application” because not only was the PGT-A industry unable to demonstrate promised outcome benefits but had to change the alleged benefits over and over again, as older alleged benefits by a small cadre of PGT-A opponents were slowly but surely and case by case disproven. This in itself was, of course remarkable because shouldn’t evidence be produced by proponents of an intervention? Instead, the opponents of an unvalidated intervention were forced to produce evidence that the intervention didn’t work.
In today’s posting we are discussing recent PGT-A related publications, some valuable, - others not so. We are, however, especially pleased to have the opportunity to present a publication that has mostly remained under the radar of general attention because it was not published in an infertility journal or a highly ranked general medical journal but in a rather peripheral general OB/GYN journal (the limited attention may, indeed, have been a purpose!).
It is a truly amazing study because it presented an acknowledgment of “defeat” by some of the most prominent proponents of PGT-A in the world, for the first time basically openly acknowledging the futility of PGT-A in association with IVF. We could not have asked for a better acknowledgment at our 20-year anniversary from having been making this point. Sometimes good things take time!
The CHR’s Editorial Staff
INTERESTING RECENT NEWS REGARDING CHROMOSOMAL TESTING OF EMBRYOS IN IVF
Any discussion of IVF between patient and physician these days – as over half of all IVF cycles in the U.S. currently utilize this test - should, of course, address the highly controversial issue of preimplantation genetic testing for aneuploidy (PGT-A), the formal name for the testing of embryos for chromosomal abnormalities prior to embryo transfer. We to this day are, however, steadily surprised how many patients reach the CHR still claiming never to have been informed about existing controversies surrounding PGT-A. The issue usually comes up when they present for an initial second opinion consultation to the CHR after often having undergone many IVF cycles with PGT-A with only few – if any – “transferrable” embryos to show for. This is, of course truly astonishing because – if correct – very obviously - at minimum - represents inadequate informed consent. The CHR, therefore strives to fill this information vacuum by steadily reporting on news regarding PGT-A and today there are, indeed, very substantial news to report.
Does PGT-A “Rescue” Poor Quality Oocytes, - Thereby Increasing the Availability of Embryos for Embryo Transfers?
This is at least what a paper of British investigators even in its headings recently suggested and – boy – does this paper not make sense!1
What the paper suggested on first impression might seem logical: Some not too great-looking blastocysts can still be euploid (of course true and not unexpected). But then the paper becomes silly because it – first – incorrectly suggested that PGT-A – by transferring only euploid embryos – lowers miscarriage rates (according to the 2024 ASRM opinion on PGT-A an incorrect statement2) and then – second – suggests that this especially will be the case in PGT-A tested blasts which, of course, is nonsense.
The need for blastocyst culture and the need for cryopreservation to do PGT-A alone, will indeed significantly reduce pregnancy chances of these embryos (especially if they look “ugly”). Add to this the high false-positive rate of PGT-A and, therefore, the decline in cumulative pregnancy chance from not transferring at least some potentially fine embryos with good pregnancy and live birth chances which erroneously have been tagged with an “aneuploidy” diagnosis (most clinics still do not transfer such embryos), and the paper’s argument of improving potential pregnancy and delivery chances makes absolutely no sense.
There is, however, another reason why we are addressing this paper her: We not too long ago in a prior posting here in quite some detail presented several arguments why editors should not publish their own papers in journals they edit. This paper is another good example for this argument because its senior author is one of the two senior editors of the journal where this paper appeared. And we know this editor well: he is a very good scientist (and editor) who has published large numbers of excellent papers. Why he really needs his name associated with this paper, therefore, also makes absolutely no sense. The connection to the next reviewed paper will also immediately become obvious.
Even Italian Colleagues Can Be Sensible About PGT-A
From its earliest days - even before being given the name PGT-A - Italian colleagues, in contrast to many other Europeans, have been among the most aggressive supporters of PGT-A.
A paper addressing PGT-A that does not push its clinical utilization is, therefore, always a pleasant surprise. Such a paper appeared recently in JARG, making the really not surprising point that – besides chromosomes – other things matter as well in human embryos.
The authors reached this conclusion after learning that, especially embryo morphology (over maternal age and developmental timing of blastocysts by imaging) wins out. And even maternal age exerts its negative effects by adversely affecting embryo competence beyond just increasing aneuploidy. In other words, they reached the correct conclusion that there must be other age-related factors in embryo physiology beyond rising aneuploidy that are the guilty parties why getting pregnant with increasingly older eggs becomes less and less successful.3
Difference in Embryo Ploidy Between Primary and Secondary Repeat Aborters -- The authors of this paper – also in JARG – addressed an interesting question,- do primary repeat aborters (women who from 1st pregnancy on miscarry) differ in ploidy (chromosomal abnormalities) of their products of conception from secondary aborters (women who had 1 or more children before starting with repeated miscarriages).
This question once again relates to reproductive immunology because – at least when it comes to immune-causes for pregnancy loss – it is well established that a first pregnancy can sensitize a mother’s immune system like a first exposure to an allergen may lead only to a mild rash, while a second exposure can lead to anaphylaxis. Here, too, the mother may have had only mild preeclampsia in her first pregnancy (or may have shown no abnormality at all) but in a second pregnancy may, suddenly, become highly symptomatic.
Such a danger may be especially profound if the first pregnancy was a male (male epitopes elicit more immune responses from mothers than female epitopes).
How Human Embryos Self-Correct
A so-called Mini Review in Human Reproduction by two British authors recently addressed the concept of self-correction in embryos downstream from blastocyst stage when trophectoderm biopsy nowadays are performed.4 And we are recommending this paper for readers interested in PGT-A not because the authors referenced four papers from the CHR but because – despite considerable evidence in support – some proponents of PGT-A are still claiming that embryos never self-correct.
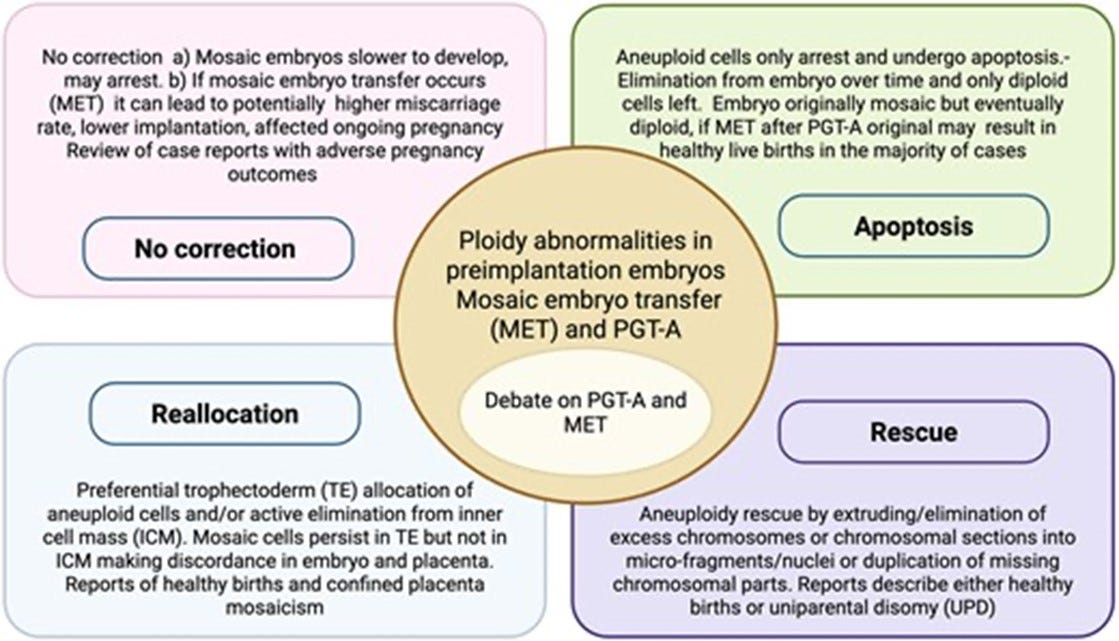
Though the authors of the review are not yet ready to commit fully to the concept of embryo self-correction, they acknowledge that embryo mosaicism is well documented in at least over 80% of preimplantation-stage embryos. And this number is likely an underestimate because without testing (almost) every cell in a blastocyst-stage embryo, - only one thing is certain: the only limited biopsy at blastocyst stage that will be 100% accurate is a biopsy that reveals mosaicism. A limited biopsy demonstrating full aneuploidy – unless meiotic in origin – (most aneuploidies, however, are mitotic) in a majority of cases still may be mosaic, - just like a fully euploid PGT-A result much more likely in reality reflects a mosaic embryo.
Moreover, with now consensus reached that aneuploid islands of cells can be found in most term placentas, segregation between euploidy and aneuploidy happens, of course, only in the embryonic cell lineage, while PGT-A biopsies are taken from the trophectoderm, - a product of the extraembryonic cell lineage. Mosaicism, therefore, represent a normal feature of preimplantation stage embryos and, indeed, may be used by embryos to avoid rejection by the maternal immune system during early stages of implantation. But that is a subject for another time!
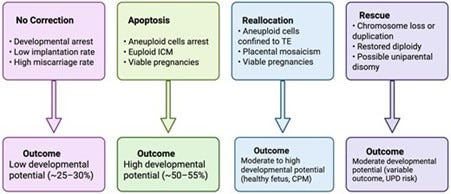
This review should leave little doubt that those arguing against self-correction still have some reading to do!
IS THIS THE PAPER THAT FINALLY SETTELS THE PGT-A DEBATE?
And then there was another paper, - astonishingly including some of the most vocal proponents of PGT-A over the last 20 years, from Dagan Wells, PhD, to Richard Scott, MD, Juan Antonio Garcia Velasco, MD, Antonio Pellicer, MD, Chaim Jalas, and Antonio Capalbo, PhD, who published - out of all journals - in the American Journal of Obstetrics and Gynecology an international multicenter study involving thousands of patients under the bombshell title:
“Preimplantation genetic testing for aneuploidy mosaicism reporting lacks clinical predictive value for live birth in a multisite, double-blinded study with independent validation” 5
And we are quoting here verbatim: The purpose of the study was to determine whether reporting putative mosaicism based on intermediate copy number improves the prediction of reproductive outcomes and should influence embryo selection in clinical practice. And the answer was that the presence of high-level intermediate copy numbers in the trophectoderm biopsy was associated with a modest reduction in live birth rate. Because of its low incidence and limited effect size, mosaic reporting, however, did not contribute to a meaningful improvement in clinical outcomes. In our clinical and laboratory setting, reporting putative mosaicism provides no clinical benefit and should not guide embryo selection in routine IVF practice.
We already noted above in discussing the preceding paper that – considering how small every trophectoderm biopsy is for PGT-A and baring a meiotic aneuploidy (which represents only as small percentage of all aneuploidies in embryos at blastocyst stage), – only a “mosaic” diagnosis by diagnosis of intermediate copy numbers can with certainty be viewed as correct. In contrast diagnoses with either seemingly “euploid” or “aneuploid” copy numbers have a high probability to actually - overall for the embryo rather than only the small 5-10-cell trophectoderm biopsy – in reality to reflect “mosaic” (i.e. intermediate copy number) embryos.
In other words, what this paper finally acknowledges is that a vast majority of PGT-A diagnoses – far beyond what currently is described as “mosaic” - lacks clinical predictive value for live births, provides no clinical benefit and should not guide embryo selection in routine IVF practice. Consequently, one has to ask the question, why are over half of all IVF cycles in the U.S. still using PGT-A?
And obviously less important but nevertheless a very important question, - one also must ask why this important paper was not published in Fertility and Sterility or Human Reproduction? Who wanted it less visible?
At the same time we, however, want to express the CHR’s appreciation to all of authors of this publication. And we do this not because this paper, of course, finally confirmed what the CHR has been trying to communicate to the infertility world for almost two decades, - but because it takes guts to acknowledge having been wrong, and this is exactly what all of this paper’s 24 authors (yes, we counted!) openly and graciously did. Thank you all!
PGT-A In Cases of Severe Male Factor?
When it rains it pours; and we, frankly, don’t know any longer whether to laugh or cry because Chinese investigators recently also published a somewhat strange paper in the quite often strange BMJ involving PGT-A which demonstrated no outcome benefits for PGT-A.6
And we – frankly – have absolutely no explanation why our Chinese colleagues chose to investigate PGT-A specifically in couples with severe male factor. At least to us, the study seems to make little sense because what does male factor potentially have to do with PGT-A? Why would or – indeed - should PGT-A affect male factor infertility?
But the Chinese colleagues, nevertheless, investigated the effects of PGT-A in severe male infertility cases by randomizing 450 couples with severe male factor infertility to ICSI with or without PGT-A. For the CHR unsurprising, PGT-A utilization did nothing regarding pregnancy and live birth rates. But – somewhat strangely and unexpected, male infertility couples who used PGT-A had lower clinical miscarriages (13/225, 5.8% vs. 43/225, 19.1%; P<0.001) as well as chemical pregnancies (7/225 vs. 3.1% vs. 19/225, 8.4%; P=0.02), - on first impression suggesting that PGT-A may have IVF outcome effects after all.
But thinking this through, positive effects on miscarriages make little sense physiologically as well as mathematically because if a statistically significant number reduction in miscarriages occurred, how come there was no concomitant increase in live births. The article presumed that PGT-A eliminated embryos prone to miscarriages but why then no increase in pregnancy, cumulative pregnancies and live births?
We smell a statistical rat somewhere in the bushes!
Severe male infertility patients demonstrated an aneuploid rate of 42.6% and - in opinion of the authors – warrants PGT-A because of these numbers. We are, frankly, not sure of that. So much more effort and cost to prevent a still questionable alleged increase in chemical pregnancies (marginal) and clinical miscarriages (statistically significant).?
SUMMARY AND CONCLUSIONS – PGT-A does not improve IVF outcomes in general populations.2 In good prognosis patients it may, however, shorten time to pregnancy and may also reduce miscarriage risk. Good prognosis patients, however, even without PGT-A have very high pregnancy and live birth rates. Routine utilization of PGT-A in good prognosis patients may, therefore, not be cost-effective.
The one still unresolved issue in PGT-A are adverse IVF effects from PGT-A utilization. More specifically, which are the subgroups of patients who not only will not benefit from PGT-A, but will actually be harmed by PGT-A. Our analysis of available data suggests that the risk for adverse outcomes is inversely related to functional ovarian reserve of patients and, therefore, to their number of available oocytes and embryos for transfer: The smaller the number, the more likely will PGT-A be harmful because the smaller the available number of embryos for transfer, the more consequential is non-use of a false-positive embryo.
REFERENCES
Al Hashimi et al., RBMOnline 2026;52(1):105208.
ASRM/SART. Fertil Steril 2024;122(3):421-433
Listorti et al., J Assist Reprod Genet 2026;43:143-153
Mantzouratou A, Mania A. Hum Reprod 2026;41(3):319-331
Gill et al, Am J Obstet Gynecol 2026;234(4):1015-1041
Lin et al., BMJ 2025;391:e084050


