NEWS ON MALE INFERTILITY
Trying to alternate for the weekend, we offer in our posting today again commentary in two distinctively different areas of clinical medicine linked to infertility: - male factor infertility in comparison to female infertility has always been the stepchild of the infertility field and, likely, will remain so, even though - as worldwide sperm counts have been declining – male factor infertility represents a growing proportion of infertility cases, with some studies suggesting that female and male infertility factors have equalized. We have, of course been as guilty as everybody else in this discrimination and, therefore, are starting today’s posting with two interesting recent publications raising some very basic questions regarding male fertility and infertility. And we then switch to pregnancy, - of course the happy outcome of every fertility treatment. Within this context, it is, however, important to remember that the ultimate goal of fertility treatments is not pregnancy but a healthy live birth. Making maximal efforts to prevent pregnancy complications before pregnancy occurs is, therefore, an integral part of infertility care and has been a very basic principle at the CHR since its establishment. Tell us what you think!
The CHR’s Editorial Staff
A Provocative Questions – Are Current Semen Analyses Still Worth It?
This is basically the question Italian investigators recently asked in a Review/Opinion article in Endocrinology1 and – maybe somewhat surprising - the answer was, - maybe not!
Here are some of the more interesting conclusions: (i) Based on WHO recommendations making semen analyses over the years more and mor complicated did not improve the ability to interpret results and to better understand “what is going on in the testes.” (ii) In association with IVF, the male partner is (within reasonable parameters), still, merely considered as a relatively unimportant “sperm provider.” (iii) Consequently, there is a general believe that there really is no need for better semen analysis tools (we very much plead guilty of holding this opinion!). (iv) Some recent investigations have suggested that sexual arousal may produce better semen samples than masturbation, - the usual way specimens are produced in fertility practice. (v) Semen analyses have been shown to differ significantly between laboratories. (vi) Some newly developed tests may have additive diagnostic value (though we are not sure of that at all!). And (vii) because of all the preceding points, the authors recommend to “move away” from the centrality of the semen analysis in evaluating men.
What this review really informed about is that the CHR’s current understanding of what a semen analysis really offers diagnostically is basically correct; As long as an infertile couple is already committed to IVF, ICSI is available, “reasonable” semen parameters regarding count, motility, and morphology, render further details in semen analyses irrelevant.
Andrologists and urologists, - please don’t hate us!
REFERENCE
Furini et al., Endocrine 2025.90:1067-1078
The Pendulum Continues Swinging When It Comes to Male Testosterone Supplementation
That androgen supplementation is not only controversial in women but may be even more controversial in men may surprise many of our readers and the principal reason is that indications are so much more variable than in the female.
Testosterone supplementation in men trying to impregnate their partners is, of course, contraindicated because it will shut down sperm production in testes. But otherwise, an FDA panel of experts recently published new recommendations for testosterone supplementation in men which significantly loosened prior restrictions.1
Comprised of urologists and health officials (but no fertility specialists), the panel recommended for men the loosening of restrictions beyond only low testosterone (T: i.e., hypogonadism), calling for expanded FDA-approved uses beyond disease-induced low T to include age-related declines. In addition, the FDA removed T’s controlled substance status. The change in recommendations was based on prior changes in guidelines from major medical societies and the observation that prior stricter rules led to more unregulated and unsupervised T utilization.1
As a panel recommendation, these changes are only “advisory” and the FDA leadership must now make final decisions, though the expectation is that this final decision will follow panel recommendations
REFERENCE
FDA. December 2025. FDA panel on testosterone replacement therapy for men. https://www.google.com/searchq=FDA+panel+on+testosterone+replacement+therapy+for+men&rlz=1C5CHFA_enUS1083US1083&oq=FDA+panel+on+testosterone+replacement+therapy+for+men&gs_lcrp=EgZjaHJvbWUyBggAEEUYOdIBCjI1NjgxajBqMTWoAgiwAgE&sourceid=chrome&ie=UTF-8
OBSTETRICAL PRACTICE AND PREGNANCY COMPLICATIONS
Most Recent U.S. Stillbirth Rates and Total Fetal Mortality
STILLBIRTHS -- The definition of a stillbirth is the death of a fetus at or after 20 weeks’ gestation. It occurs annually in over 20 000 pregnancies in the U.S. alone, representing a rate of 5.74 per 1000 US births (10.34 per 1000 births among non-Hispanic Black individuals. Approximately half of at least stillbirths at term are believed to be preventable, - yet little progress has been made in reducing stillbirth rates. In 2023, the Department of Health and Human Services, therefore, established a Stillbirth Working Group.
Harvard investigators now reported in JAMA on singleton births between 2016 and 2022 on unadjusted prospective fetal mortality rates overall and in a subgroups of third-trimester births among health insurance claims. Among 2,792,699 total births, 18, 893 (6.8/1,000 births) were recorded. Mortality rates were ca. 0.7/1,000 at 20 weeks, steadily declined to a nadir of 0.13/1,000 at 29 weeks, only to rise back up to similar numbers as at 20 weeks by 38 weeks, - and then quickly rising further to an all-time peak of 2.29/1,000 above 41 weeks.
Interestingly, these numbers were higher than 2021 rates reported by the CDC. In addition no risk factors were identified in 27% of all stillbirths, and in 40.5% of stillbirths even above 41 weeks gestational age, which led the investigators to suggest that we must get better in risk stratifying.
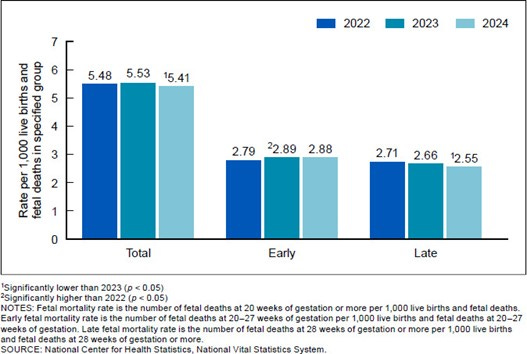
And obviously related, the CDC reported fetal mortality in the U.S. with final numbers for 2022-2023 and provisional numbers for 2024.2 Comparisons showed differences between 2022 and 2023.
FETAL MORTALITY -- From 2023 to 2024, the overall fetal mortality rate declined 2%, from 5.53 to 5.41 fetal deaths at 20 weeks of gestation or more per 1,000 live births and fetal deaths. During this time, the early fetal mortality rate (20–27 weeks of gestation) was essentially unchanged (2.88 in 2024), while the late fetal mortality rate (28 weeks of gestation or more) declined 4% (2.55 in 2024, see Figure below).
No significant changes in fetal mortality rates between 2023 and 2024 were observed among the race and Hispanic-origin groups. Fetal mortality rates decreased in 3 states, increased in 1 state, and were not significantly different in 46 states and the District of Columbia from 2023 to 2024. In comparison, from 2022 to 2023, the fetal mortality rate increased for early fetal deaths, for Asian non-Hispanic women, and in five states, and declined in one state (see Figure below).
This analysis of provisional 2024 fetal mortality data found that the total and late fetal mortality rates declined by 2% and 4%, respectively, compared with 2023 rates, while the early fetal mortality rate was essentially unchanged. From 2023 to 2024, no significant changes were observed in the fetal mortality rates for any of the race and Hispanic-origin groups. In comparison, from 2022 to 2023, the fetal mortality rate increased for early fetal deaths, for Asian women.
REFERENCES
Sullivan et al. JAMA 2025;334(22):2033-2035
Gregory et al., CDC. NCHS, National Vital Statistics System. December 2025. Report No 41. https://www.ncbi.nlm.nih.gov/books/NBK619812/
Pregnancy As an Accelerated Aging Event for Women
That pregnancy can accelerate female aging has over the last year been a subject that attracted considerable attention in the medical literature. We, therefore, previously addressed the subject in these pages but - as Obstetrics & Gynecology recently once more contributed two articles to the discussion - we felt that another round of comments was indicated.
In the first paper colleagues from Stanford University investigated whether longitudinal epigenetic maternal aging in pregnancy was associated with adverse pregnancy outcomes.1 These data were obtained with 11 epigenetic clocks from Illumina EPIC 2 arrays, - one of them being GrimAge2. Based on earlier reports, what they found was not unexpected: Pregnancy, indeed, accelerated epigenetic aging within a nulliparous woman by up to 5.3 years.,
Older first trimester epigenetic age by GrimAge2 (but not chronologic age) was marginally (p=0.04) associated with a composite of pregnancy complications which – interestingly – were mostly immune system-related, considering the CHR’s longstanding interest in immunology of course a very interesting finding for the CHR’s investigators. Considering that older women currently represent the only remaining age group with still increasing pregnancy numbers, the authors offered the very astute conclusion that
epigenetic clocks should be used in such patients to investigate their risks for pregnancy complications in the first trimester of pregnancy. In the CHR’s opinion, such investigations in older women, could= and should -, indeed, already start during infertility care.
Two colleagues from Columbia University, among them Zev Williams, MD, PhD, Chief of REI at Columbia then contributed a commentary and editorial to the same issue of Obstetrics & Gynecology, correctly noting that – if confirmed – these findings may require a “reconceptualization” of what defines “advanced maternal age.”2 They also pointed out that these findings are supportive of the longstanding concept of pregnancy as a “stress test” for practically all organs and bodily systems of a woman. Both papers are important contributions.
REFERENCES
Panelli et al., Obstet Gyecol 2025;146(6):783792
Williams Z, Suh Y. Obstet Gynecol 2025;146(6):781-782
The T Cell That Likely Helps in Maintaining a Healthy Pregnancy (and also Protects from Autoimmunity)
Here is yet another story regarding reproductive immunology – obviously a topic we, here at the CHR cannot get enough of – that demonstrates how closely intertwined pregnancy-tolerance is with self-tolerance, - i.e., how much in common autoimmune conditions have with certain immunological pregnancy complications.
The subject is the KIR+ CD8+ T cell, and the story starts with a paper by investigators from the University of Pittsburgh (Li et al1) in Science Translational Medicine which an editor summarized (this journal smartly starts each paper with an Editor’s Summary).
And this is how she summarized this important paper: In pregnancy the maternal immune system must tolerate the usually semi-allogeneic fetus (in cases of egg donation and in gestational carrier pregnancies the fetus is now fully allogeneic), while still maintaining sufficient immunity to continue reasonably protecting the mother from infections. The investigators in this paper now reported that certain CD8+ T cells expressing killer cell immunoglobulin-like receptors (KIRs) – an only relatively recently identified regulatory CD8+ T cell population - are in peripheral blood and placental tissue enriched during human pregnancy and express ex-vivo very potent suppressive activity.
As Asher Jones, a science communicator and writer at the University of Pittsburgh noted in a recent communication from the Department of Immunology, KIR+ CD8+ T cells were higher in pregnant people and increased during the second trimester. 2 Moreover, as one, therefore, would expect (because of their Y-antigens), male fetuses produced higher levels of KIR+ CD8+ T cells than female babies. The researchers, then went into in vitro studies, - taking T cells from mothers and combined them in a dish with umbilical cord cells from their babies. When KIR= CD8+ T cells were removed, the mothers’ other T cells reacted much more strongly to the fetal cells, demonstrating their critical role in suppressing maternal immune responses against the fetus.
In pregnant with likely immunologically-induced miscarriages and/or preeclampsia these KIR+ CD8+ T cells were found further enriched above normal pregnancy levels, suggesting that they may try – unsuccessfully – to control inflammation but were failing. These findings, however, nevertheless suggested that these cells play an important role in maintaining maternal immune balance in normal pregnancy. The authors then also correctly noted that that – due to their functions - they may become candidate biomarkers or even therapeutic targets in pregnancy complications like repeated immune-mediated pregnancy loss and preeclampsia.
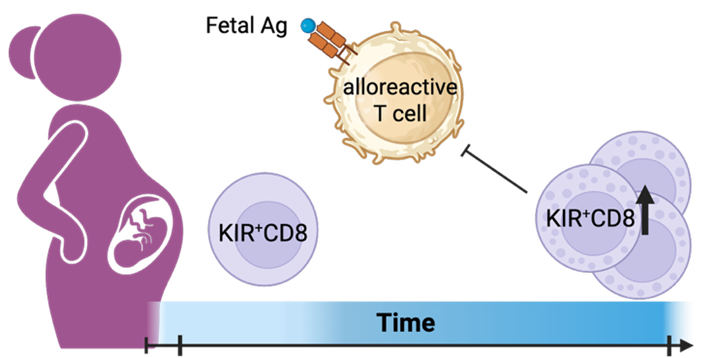
As Li et al (then still at Stanford University) already in 2022 reported, these cells have also been demonstrated to be active in autoimmune diseases, such as celiac disease, multiple sclerosis, and systemic lupus erythematosus, as well as with COVID-19,3 of course closing for us here at the CHR the unifying circle between pregnancy tolerance and self-tolerance we referred to at the beginning of this commentary.
REFERENCES
Li et al., Science Tranlat Med 2025;17(810). DOI: 10.1126/scitranslmed.adm7697
Jones A. Department of Immunology. University of Pittsburgh. October 13. 2025. https://www.immunology.pitt.edu/news/meet-t-cells-help-maintain-healthy-pregnancy
Li et al., Science 2022;376(6590):eabi9591
New Information About the Development of the Fetal Immune System
In a quite remarkable paper in Cell, Chinese investigators recently offered new information about the developing fetal immune system, especially during the second trimester of pregnancy, a pivotal stage in human immune system development (and a time when maternal tolerance toward the fetal semi-allograft is at its peak). Here is some detail: Utilizing single-cell RNA sequencing and T cell receptor sequencing, the authors profiled 2,868,420 immune cells from 321 samples across 23 fetal organs, using adult tissues for comparison, identifying an extrathymic CD4+ T cell subset mediating TOX2+ precursor cells’ transition to mature naive CD4+ T cells.
Contrary to the widely held current believe of fetal immune quiescence, they uncovered widespread memory -activated T cells and tissue-resident memory clones shared across organs, indicating systemic immune activity beyond localized barrier defense. Cell-cell communication and functional assays indicated two tolerance mechanisms that suppress fetal T cell activation: ARG1+ neutrophils and a PTGES3/PTGER4 signaling pathway. They also found that hematopoietic stem cells (HSCs) disperse across multiple organs and demonstrated that HSCs from non-canonical hematopoietic organs differentiated into diverse immune lineages (see graphic abstract below).
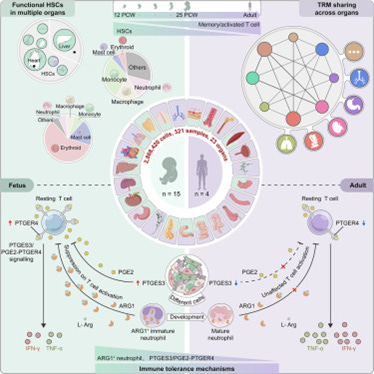
REFERENCE
He et al., Cell 2025; 188(25):P7291-7308.E23
Does Hydroxychloroquine (Plaquenil) Treatment in Pregnancy Help in the Anti-phospholipid Syndrome (APS)?
As just noted above, here at the CHR researchers for decades have had exceptional love for the commonalities between pregnancy- and self-tolerance. Yet in parallel, we also developed a clear dislike for what our rheumatology colleagues have originally come to call the anti-phospholipid syndrome (APS). The syndrome, however, not only changed over the years definitions, - but also names. And while there were several reasons for our dislike of what our rheumatology called the APS, the principal one for the CHR has been that we see this alleged diagnosis as “concocted.”
Its history started many decades ago in New Zealand when an internal medicine practitioner in a hospital ward coincidentally noted several women who just had experienced miscarriages, had deep vein thromboses, and were found to be positive for the so-called lupus anticoagulant (LA). Because of the unusual confluence of patients with identical presentation, this practitioner then reported this case series in The Lancet.1
It turned out to be an important new observation, and the CHR’s Norbert Gleicher, MD, was, indeed, among the first – if not the first - to follow up on it with an article in JAMA, pointing out that the syndrome was not only defined by a positive LA but also by typical autoimmune autoantibodies.2
But then our rheumatology colleagues took over and established the so-called anti-phospholipid antibody syndrome (APAS) as an independent clinical diagnosis, initially defined as a woman with positive LA, a history of thrombosis, and repeated (at least 2) miscarriages. And that never made sense to us here at the CHR because without pregnancies there, of course, are no miscarriages. The diagnosis of APAS, therefore, could not be reached unless a woman experienced at least two miscarriages. What kind of diagnosis is this, when one must wait for at least two miscarriages before being able to fulfill diagnostic criteria of diagnosis. What about women who never got pregnant? And what about men?
Unsurprisingly, names as well as diagnostic phenotypes changed, but never to a degree that here at the CHR made us more comfortable with this diagnosis, - especially as it related to pregnancy. But this, of course, also did not mean that we ignored the topic when it was addressed in the medical literature. To the contrary, like anything autoimmunity-related, we followed the subject with great interest and, therefore, here are presenting a newly published study by mostly Israeli investigators. In a so-called Systematic Review and meta-analysis, they attempted to determine whether hydroxychloroquine use in APS represented effective treatment.3
By way of background, it is generally believed that women with APS have higher pregnancy risks and that roughly 20% of them - despite supposedly appropriate treatment – will still miscarry. Our rheumatology colleagues love the anti-inflammatory hydroxychloroquine (Plaquenil) as a treatment in pregnancy for several autoimmune conditions, including APS, arguing that it offers – especially in regard to miscarriage risk - outcome improvements. Through a Systematic Review and meta-analysis of the literature they now
claimed to have confirmed the positive outcome effects of the medication in pregnancy by increasing live birth rates significantly (i.e., reducing miscarriage risks) and lowering obstetrical risk in general.
To say it mildly, - we were and still are quite skeptical!
While acknowledging our well-known bias against Systematic Reviews with meta-analyses (in most cases basically only confirming the old IBM dictum. - “garbage in, garbage out”), the authors’ circular thinking pattern even further enhanced the dictum in this case and is deserving of further comments: The paper noted in the Introduction section that in APS, - LA, anti-cardiolipin (ACAs) and anti-beta-2-glycoprotein-1 antibodies , previous pregnancy loss, a history of thrombosis, and a history of systemic lupus erythematosus (SLE) are all associated with increased risk for adverse pregnancy outcomes.
In other words – as noted earlier – if you haven’t had pregnancy losses, you don’t qualify, - but if you have SLE you do qualify. But now arises the question, how does one differentiate between SLE and APS causing adverse outcomes? And why is everybody just looking only at ACAs and anti-beta-2-antibodies among anti-phospholipid antibodies (APAs)? The truth is unfortunately not very comforting: they in most laboratories are the only APAs tested.
In other words, studies on ACA basically never have a uniformly defined patient population. As readers of the CHR’s publications by now of course know, - this turns every meta-analysis into a crapshoot!
And since we are already talking about crapshoot, the meta-analysis included only seven retroactive (!) cohort studies and not even a single prospective cohort study (one these days, of course, can only dream about prospectively randomized studies in the fertility field). In short, this study would make a very good case study on how not to do Systematic Reviews with meta-analyses and, therefore, very clearly deserved critique when previously discussed in the CHRVOIVCE.
This, of course, does not mean that hydroxychloroquine/Plaquenil may not have benefits in the treatment of certain autoimmune diseases; but let’s be honest about what we do and do not know! And to our rheumatology colleagues, - we still don’t like the concept of the APS!
REFERENCES
Lubbe et al., Lancet 1983;321(8338):P1361-1363
Gleicher N, Friberg J. JAMA 1985;253:3278-3281
Berman et al., Rheum Musculoscelet Dis Open 2025;11(3):e005825.