THE INFERTILITY DIAGNOSES BY THE PUBLIC CONSIDERED MOST DIFFICULT TO TREAT
By the Editorial Team of the CHR, - which can be contacted though the editorial office of The Reproductive Times or the CHRVOICE.
In today’s posting, we attempt to primarily address infertile patients in our in interests quite diverse readership. Yes, - we got your message that you want more simple, clinical attention and we, therefore, decided to ask several A.I. platforms what the subjects are you most worry about because you consider them to be the most difficult ones to treat.
And what we, therefore, today are posting are brief summaries of the subjects the A.I. world directed us toward with special emphasis on issues where the CHR disagrees with what you may be hearing elsewhere.
By doing so, the CHR, of course, does not want to imply that the CHR is always right and others are always wrong (even though - at least in most such cases - that is, of course, the case), but we want to reemphasize that there are difference of opinion within the infertility field and the CHR feels that patients are frequently not advised of these controversies.
So, for example, we are still almost daily reminded by patients that many fertility clinics to this day fail to inform their patients about the controversies surrounding preimplantation genetic testing for aneuploidy (PGT-A), all-freeze cycles, etc. Franky, - we consider this not only surprising, - but shameful because it denies patients self-determination.
We hope you will enjoy today’s posting, and we promise to address the direct needs of our non-medical readership more often. What today’s posting, however, also reveals is that we read your comments and take them seriously. So continue sending them!
The CHR’s Editorial Staff
Like hopefully most providers of fertility services, the CHR is always interested in improving its institutional understanding of how the public views various fertility services routinely provided by most fertility clinics in importance complexity levels, accessibility, etc. We, therefore, recently asked several A.I. platforms which infertility diagnoses the public perceives as most difficult to treat. And – though there were small discrepancies between various platforms, there was surprisingly considerable unanimity and we, therefore, decided to summarize these subjects for this issue of The Reproductive Times.
It seems that everybody agrees that a single universally agreed upon “hardest” infertility problem does not exist, but in clinical reproductive medicine a few conditions consistently stand out as the most difficult to treat and least responsive to current therapies. The answer also depends on how “difficult” is defined. It ultimately depends on whether “hardest” means biologically untreatable, low success rates, or limited available interventions. Here are the main contenders:
Diminished Ovarian Reserve (DOR) / Ovarian Aging, at the CHR Called Low Functional Reserved (LFOR)
Because the CHR’s worldwide reputation as a leading fertility center of last resort evolved over the years to a large degree from the center’s innovative treatment of what in the infertility literature is usually called diminished ovarian reserve (DOR) – though the CHR prefers the term low functional ovarian reserve (LFOR) – we were very pleased to learn that most platforms considered DOR/LFOR the most fundamentally limiting problem in infertility care.
Here is why the CHR prefers the term LFOR: Everybody agrees that the total ovarian reserve is made up of two components, - with the first being the so-called pool of resting (or primordial) follicles – and the second pool being so-called growing follicles (i.e., the follicles that have been recruited out of resting stage and over three or more months either grown and mature toward ovulation or die and get absorbed).
When a patient’s ovarian reserve is evaluated with either FSH and/or AMH measurements (or even antral follicle count) we are, however, only evaluating the growing (i.e., functional) follicle pool. We have really no way to assess the resting follicle pool size, except for the fact that resting and growing follicle pools maintain a correlation in their respective numbers: A small growing follicle pool, for example, also means a small resting follicle pool and, of course, vice versa. Figure 1 below demonstrates the decline in growing follicles with advancing female age and the in parallel increase in poor quality oocytes produced by those remaining follicles in ovaries with advancing female age.
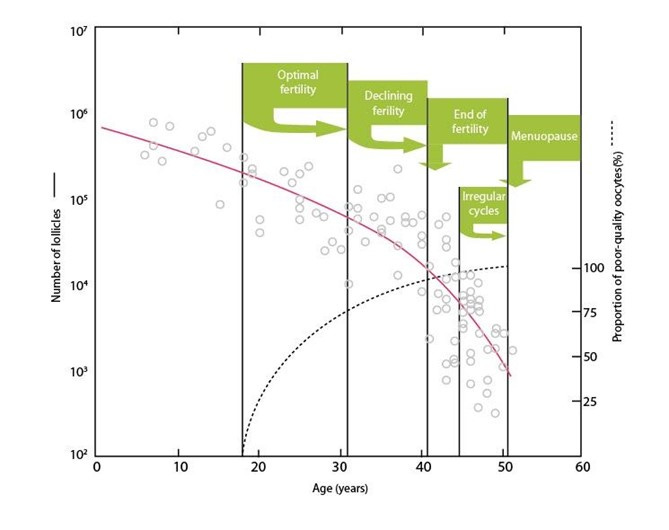
Since follicles produce the AMH hormone, its levels reflect the FOR. Approximately 10% of all women -independent of race, ethnic background or any other patient characteristic will end up with a diagnosis of premature ovarian aging (POA) which, of course, can be mild, moderate or severe. Approximately 1% of those 10% (i.e., 1 in 100) will demonstrate primary ovarian insufficiency (POI) defined by amenorrhea, FSH above 40.0 mIU /mL, undetectable AMH, - all reached before age 40.
So why is treating DOR/LFOR so relatively difficult?
The ovary contains a finite, non-renewable pool of follicles.
Both quantity and quality (aneuploidy rates) decline with age.
Pregnancy chances are strongly tied to egg biology, which currently can still not be reversed.
Clinical reality is that:
Even with in vitro fertilization (IVF), success rates drop sharply with age.
No therapy reliably improves egg quality in everybody, though some therapies are effective in selected patient groups.
Egg donation is, therefore, often the only highly effective option, which – unfortunately – leas many, if not most, IVF clinics to conclude that all women above age 42-43 should be advanced into egg-donation cycle.
Increasing numbers of fertility clinics leave patients, indeed, no choice.
This is, however, not the CHR’s practice: At the CHR, patients are not told how to live their lives. They instead are – with full transparency – told their chances with all available treatment options, - and then it is the patients’ choice which treatment they wish to receive.
It is, likely, the strong desire of most patients (husbands often even more so than their wives to – first of all - work with their own eggs, - even if that means lower pregnancy chances than with donor eggs.
Donor eggs can be and are nowadays frequently used into the mid-50s (and sometimes beyond); they, therefore, remain an option for quite some time, while a woman’s own eggs, of course, vanish.
Poor Oocyte and - Consequently - Embryo Quality Due to Various Causes Including Aneuploidy
Oocyte and, therefore, embryo quality are obviously linked to aging; but they, nevertheless, can be conceptually distinct in what the cause is for poorer quality. The best way to illustrate this fact is to point out that there are young women with older behaving ovaries and older women with young behaving ovaries, - just as there are young women who look older than they are and older women who look younger.
Which brings us to what makes and oocyte be of poor quality? And the answer is that advanced age, can, of course, be responsible; but, even if poor quality is age-related, it can have several different causes. It may at random be a “bad” cycle; the patient may have received inappropriate gonadotropin stimulation; the timing of her egg retrieval may have been off; the embryology laboratory may not be great, and once egg quality is bad, embryo quality will be bad as well.
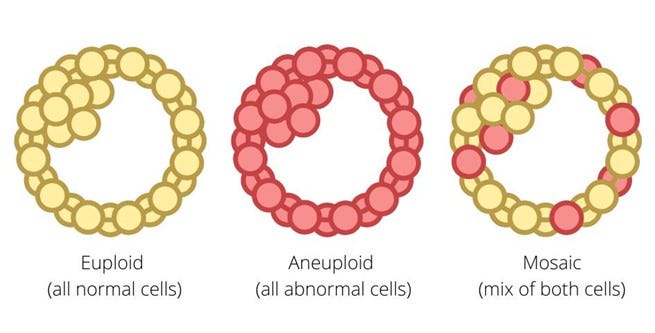
And then there is, of course, aneuploidy (an abnormal set of chromosomes), - with the likelihood of aneuploidy in oocytes and embryos increasing with age. Figure 2 below schematically demonstrates how a blastocyst-stage embryo is diagnosed in what is called preimplantation genetic testing for aneuploidy (PGT-A) resulting in either a “normal’ (i.e., euploid) test result, in “aneuploidy” (all cells presumed to be aneuploid), and in a “mosaic” result (the embryo is presumed to have a mix of normal and abnormal cells.
Because miscarriages in over half of all cases were demonstrated to show abnormal karyotypes (abnormal chromosome numbers), the hypothesis arose over 20 years ago that IVF pregnancy and live birth rates (per embryo transfer) could be improved by testing an embryo’s chromosomal complement before transfer, - and transferring then, therefore, only “euploid’ embryos. This testing, initially given the name preimplantation genetic screening (PGS) or preimplantation genetic diagnosis (PGD) was later renamed PGT-A.
Investigators at the CHR – after reanalyzing published data form Belgian investigators, - however already in 2005/2006 developed serious concerns about the hypothesis of testing embryos in this way and not only concluded that this kind of testing very likely did not improve IVF cycle outcomes but – especially in older women, indeed, reduced pregnancy chances. Almost 20 years later, even the American Society for Reproductive Medicine (ASRM) finally concluded in September of 2024 in a formal “Opinion” that PGT-A to this day has not demonstrated any outcome benefits for IVF, - but to this day has still not made a final statement concluding – as the CHR’s investigators and a small group of other scientists by now have well established – that PGT-A utilization for several subpopulations of IVF patients, especially women who produce small embryo numbers and/or who are older – may, indeed, reduce pregnancy and live birth chances.
There are many reasons for this and here is not the place to go into further detail (see our Reading List for that purpose below) but it is now really already appropriate to say that PGT-A is simply a “lousy” test which for several reasons never can offer a reasonably accurate embryo diagnosis about its ploidy status to make the crucially important decision whether an embryo deserves to be excluded from transfer. Since most errors in diagnosis are so-called “false-positive” diagnoses, this means that such a diagnosis automatically reduces a patient’s cumulative pregnancy and live birth chance which this falsely deselected embryo usually would have offered the patient.
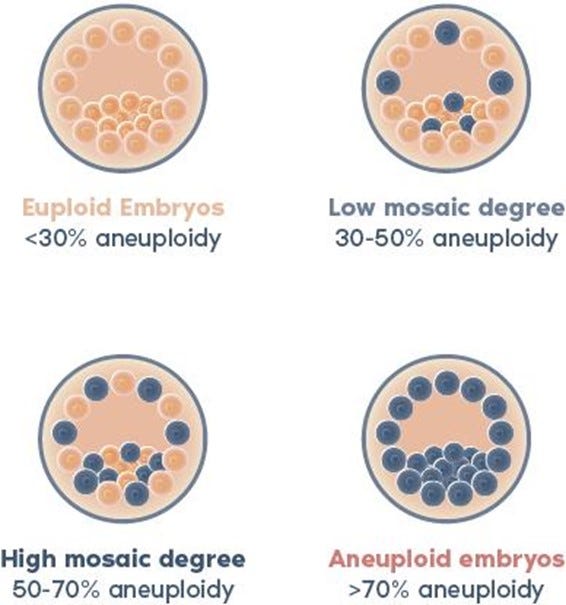
The genetic testing laboratory industry in 2016 only further confused the issue by introducing the diagnosis of “mosaicism” to PGT-A (before that date embryos were diagnosed only binary, as either “euploid” or “aneuploid.” Even though simple logic would already suggest that taking at random from a ca. 250 cell blastocyst-stage embryo a biopsy of only on average 5-6 cells mathematically cannot reflect the complete embryo, the introduction of “mosaicism,” of course only further complicated, therefore, the interpretation of results. Figure 3 below schematically demonstrates the concept.
In contrast to most other IVF clinics in the U.S. and really the rest of the world, the CHR, therefore, does not recommend the routine utilization of PGT-A in IVF cycles, though, of course offers the test if patients so desire. The test is unfortunately too often described as “accurate” in diagnosis, though “unfortunately unable to correct those embryos.” This is a categorically incorrect description of the test. The principal shortcoming of PGT-A does not lie in its inability to “correct” chromosomal abnormal embryos but in the test’s very high false-positive diagnosis rate which leads to disposal or other non-use of many embryos with good pregnancy and delivery chances for chromosomal-normal offspring.
This was also proven for the first time by the CHR where investigators (in collaboration with two at that time still existing other IVF clinics) in 2014 started transferring selected by testing as “aneuploid” diagnosed and reported embryos. In the fall of 2015 the CHR reported the first 4 chromosomal-normal pregnancies after transfer of such “abnormal” embryos. The CHR has continued this practice and established a case registry for such transfers, which every few years is published, demonstrating a steadily growing number of healthy children born from those transfers.
Since the CHR, as noted above, does not recommend PGT-A to its own patients, most of the “abnormal” embryos the center transfers are moved in from other IVF clinics which – to this day – refuse such transfers.
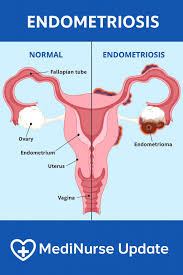
Severe Endometriosis
The emphasis here is on the word “severe” because – frankly speaking – nobody even knows with certainty what the prevalence of mild endometriosis really is. After all, a significant portion of mild endometriosis is microscopic. Even laparoscopy, therefore, can miss endometriosis. We, therefore, here do not offer a general discussion of endometriosis in regard to infertility but will try to make a few important points which are not often well understood by infertility patients and the public in general.
• Not every woman with endometriosis is automatically infertile. Indeed, many women with endometriosis are normally fertile and may not even know that they have endometriosis.
• Mild endometriosis can be very symptomatic, while even very severe endometriosis may be so asymptomatic that patients don’t even suspect that they have the disease.
• Once a decision has been reached that a patient requires IVF, whether she does or does not have endometriosis no longer matters because presence vs. absence of endometriosis does not change IVF protocols.
• Therefore a diagnostic laparoscopy after one or more failed IVF cycles “to find out whether a patient has endometriosis – as we frequently see in the history of patients – makes absolutely no sense.
• All of this also means that so-called restorative reproductive medicine (RRM), proposed by some right wing political pundits in a Republican think tank, supposedly meant to treat “root causes” of infertility like endometriosis (through surgery) rather than IVF, is pure nonsense.
•. There are good reasons why IVF has replaced surgery in most cases of endometriosis when the goal is fertility. Surgery may, however, still be the best treatment choice when the primary problem is not infertility but pain.
•. And a final word about the use of surgery in endometriosis: It can be very difficult to say “no” to cutting away once one is in surgery and sees pathology. Especially in the older days it was not uncommon to see women going into endometriosis surgery with good ovarian reserve and coming out of surgery in menopause.
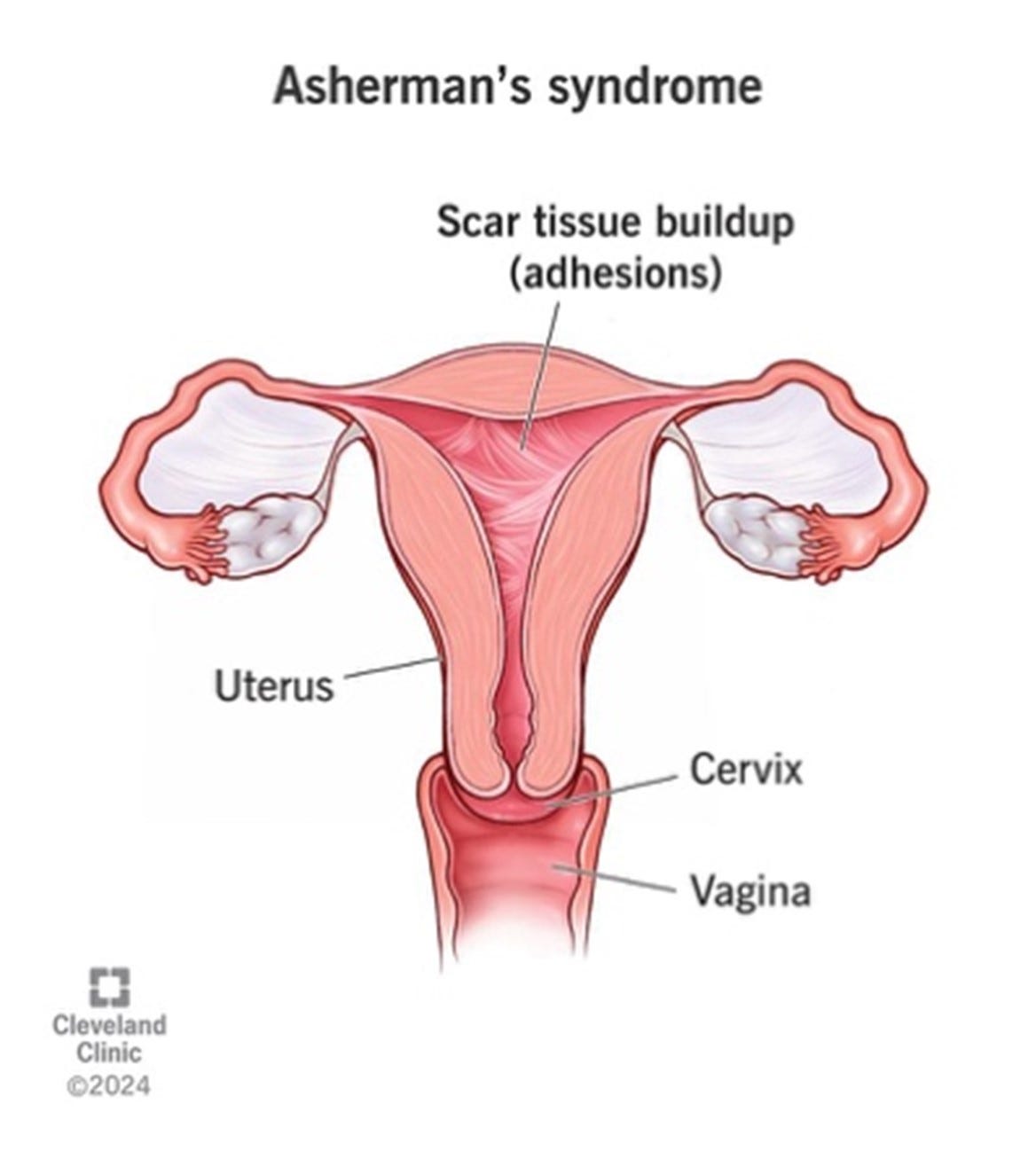
From Intrauterine Adhesions to Full Blown Asherman’s Syndrome
That this subject would rank among the most difficult to treat fertility related conditions came somewhat as a surprise, - not because a true Asherman’s syndrome (see Figure 5) obliterating part or all of an endometrial cavity is not, indeed, an extremely difficult to treat condition but because true Asherman’s are fortunately rare. In contrast, endometrial adhesions here and there are quite frequent, - but in most cases do not represent a major treatment problem.
Some very recent reports from Spanish investigators suggested that bone marrow-derived stem cells may turn out to improve standard treatments of Asherman’s syndrome which traditionally only involved dissection of the scar tissue during hysteroscopy with insertion of a ballon into the endometrial cavity to prevent re-adhesion of anterior and posterior uterine walls and accompanied by high-dose estrogen treatments.
Severe Uterine Factor Infertility
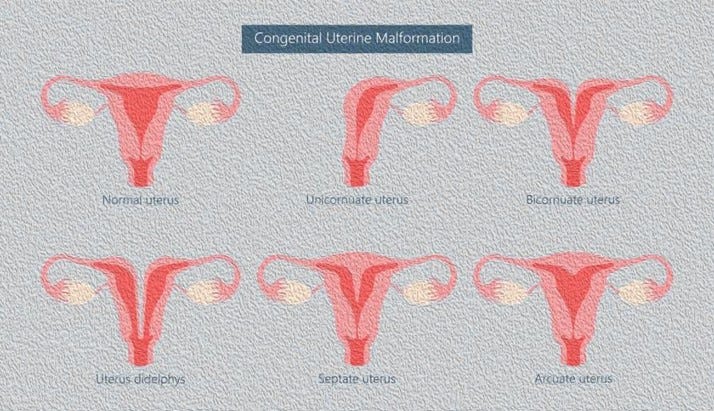
Once again the emphasis is on the word “severe” because minor uterine pathologies are usually not too difficult to overcome. Figure 6 offers schematic drawings of congenital – so-called Müllerian uterine abnormalities. Most of these abnormalities are best left alone but the literature suggests that so-called septate uteri may benefit from septum resections. One has, however, to be careful in differentiating between septate and arcuate uteri, because the latter does not benefit from resection and risks uterine perforation.
Uterine reconstructions for bicornuate or even diadelphy uteri, in contrast, are usually major surgeries, requiring special surgical skills. The same applies to reconstruction of uteri after major myomectomies because myomas (fibroids) – especially if protruding into the endometrial cavity can interfere with implantation and/or increase miscarriage risk if embryos implant on such a protruding fibroid.
“Unexplained” Infertility also Called Idiopathic Infertility
This is a frustrating diagnosis for patients as well as physicians because it basically states that the treating physician has no idea what a patient’s (infertility) diagnosis is. For many really good reasons, the CHR has, indeed, very “strong” (negative) feelings about this diagnosis, which in many textbooks is still considered a valid infertility diagnosis. Indeed, some colleagues claim that it may involve up to approximately one-third of all infertility cases.
We don’t think so and, indeed, consider this diagnosis an outright oxymoron. And here is why: Like in any attempt in life to discover something, the success of the efforts will depend on how deep one digs. Infertility is not different: An attempt at finding one or more causes for a couple’s infertility can be more or less superficial (or should we say more and less in depth). Several years ago, the CHR’s investigators, indeed, published several papers on the subject, arguing that – if the investigation is only deep enough – this diagnosis basically does not exist.
This is also a very basic reason why obtaining a very detailed patient history and then performing – based on this past medical history – a very detailed and directed diagnostic work-up is at the CHR considered of major importance. In short, we do not believe in “unexplained” infertility!
Repeated Implantation Failure (RIF)
This is yet another widely applied diagnosis in the fertility field, the CHR has significant problems with. And once again, here is why: The medical literature in general considers a patient to qualify for the diagnosis of RIF after two unsuccessful attempts at embryo transfers in IVF with good quality embryos. And this definition – to say it mildly – is absurd because even at peak fertility (ca. at age 25) only 1 in 3 good quality embryos will lead to pregnancy.
How do we know this? Because it takes a couple at peak fertility on average ca.3.4 months to conceive. And since every normal couple at that age can be expected to produce on average one embryos every month, the math is simple and straight forward. As women get older, their oocytes (eggs) become progressively less efficient in producing good quality embryos and, therefore, pregnancy chances decline. Though numbers are more discrepant by age 45 than age 25, it is reasonable to assume that by that female age the 1:3 ratio of embryos leading to pregnancy reaches a ratio of 1:15 to 1:20.
To consider two failed embryo transfers with good quality embryos as qualifying for a RIF diagnosis, therefore, not only is not sensical for a couple at peak fertility around age 25, - but makes even progressively less sense as women advance in age. In other words, to reach a RIF diagnosis independent of considering a patient’s age is – as noted above - simply absurd and is just another example for how often too much of currently widely accepted infertility practice makes absolutely no sense!
Suffice it to say, the CHR never makes a RIF diagnosis without adjusting expectations to female age which in practical terms means that even in the CHR’s highly adversely selected patient population - which frequently involves women who failed many IVF cycles even before coming to CHR – we hardly ever end up considering a patient a RIF patient.
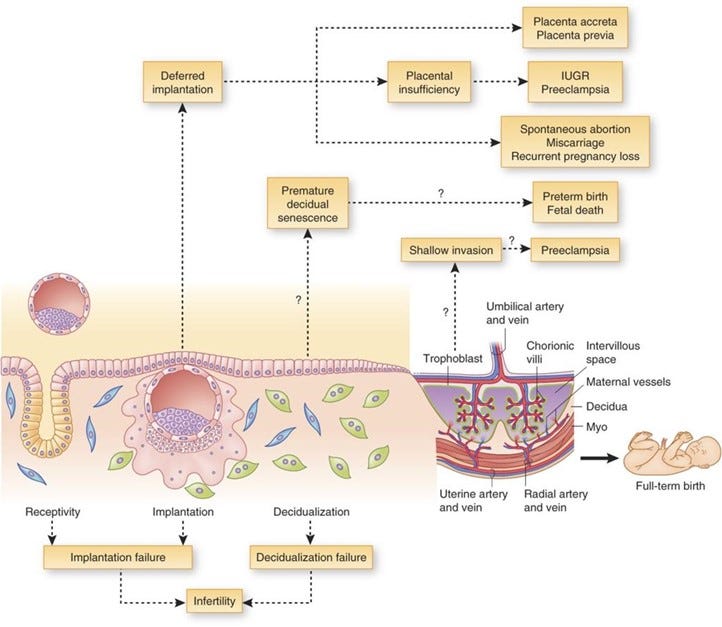
And why is this important? Figure 7 below offers the answer: The figure not only depicts graphically possible ways to real IF but also the possible obstetrical and neonatal consequences one has to consider from such a diagnosis. In other words, a false-positive diagnosis of RIF will lead to a considerable number of unnecessary and costly treatments and, in the end, may indeed negatively affect outcomes.
Immunological Infertility Including Repeated Immunological Pregnancy Loss
Because the CHR, since its founding, has been one of only very few fertility clinics not only practicing reproductive immunology but conduction an ongoing research program in reproductive immunology, this has been a subject of repeated and constant discussion in these pages. We, therefore, will not go into much detail but, still, want to reiterate a few key points where the CHR – once more – disagrees with what likely represents a significant majority of colleagues in the field:
• Because pregnancy under normal circumstance is a semi-allograft (50% genetically “foreign” transplant from the male partner) and a full (100%) allogeneic transplant in cases of egg donation, gestational carrier pregnancy or embryo donation, the CHR considers pregnancy in principle to be primarily an immune system-driven condition, - as tolerance induction in the maternal immune system must predate any other maternal influence on the pregnancy, - including endocrine effects.
• Most colleagues disagree with this conclusion and, therefore, still view pregnancy primarily as an endocrine condition.
• Considering this divergence of opinion, it cannot surprise that the CHR considers immunology as an essential component of infertility practice, - while a majority of colleagues consider immunology at best as a minor influence and, often, as completely irrelevant.
• In some contradiction, even immune-skeptic colleagues, however, usually acknowledge that immunology plays a role in implantation and in some pregnancy losses.
• The CHR believes that involvement of the maternal immune system in (a still unknown percentage of) miscarriages must be considered as established, with this kind of pregnancy loss being called immunological pregnancy loss.
• Because the ultimate role of the maternal immune system in the implantation process is still only incompletely understood, many aspects of this immune response (normal or abnormal) are becoming increasingly clear, - among those that establishment of tolerance likely is not a one-step process that either functions normally or not. It instead likely involves several steps over time, starting with local immune tolerance followed by systemic tolerance, - likely fully completed only by the beginning of the second trimester of pregnancy.
• How the maternal immune system, ultimately, tolerates the logarhythmically growing “tumor of pregnancy” is still not well understood (organ transplantation demonstrates how difficult adequate tolerance development is even if the transplant – except for liver transplants – usually does not increase in size). Some evidence, however, suggests that this may be a so-far undefined function of the to the fetus extracorporeal placenta and its maintained aneuploidy environment.
Consequently, patients with reproductive immune problems can expect at the CHR a very different treatment approach than at most other IVF clinics.
READING LIST
Gleicher N, Weghofer A, Barad D. Defining ovarian reserve to better understand ovarian aging. Reprod Biol Endocrinol 2011; 9:23
Cohen J, Chabbert-Buffet N, Darai E. Diminished ovarian reserve, premature ovarian failure, poor ovarian responder – a plea for universal definitions. J Assist Reprod Genet 2015;32:1709-1712
Gleicher N, Barad DH, Patrizio P, Orvieto R. We have reached a ded end for preimplantation genetic testing for aneuploidy. Hum Reprod 2022;37(12):2730-2734
Orvieto R. Preimplantaton genetic screening – the required RCT that has not yet been carried out. Reprod Biol Endocrinol 2016;14:35
Zondervan KT, Becker CM, Missmer SA. Endometriosis. N Engl J Med 2020;382:1244-1256
Santamaria X, Pardo- Figuerez, Gonzalez-Fernandez , Querol S, Rodruguez L, Valcarcel D, et al., Autologous cell therapy with CD133+ bone marrow derived stem cells for Asherman Syndrome a phase 1&2 trial. Nat Commun 2026;17:1093
Santamaria X, Rosen B, Perez-Moraga R, Verkatesan N, Pardo-Figuerez M, et al., Nat Commun 2023;5890:14
Gleicher N, Barad D. Unexplained infertility: Does it really exist? Hum Reprod 2006;21(8):1951-1955
Kricinska AM, Maksym RB, Szewcyk G. Immunological causes of Infertility: Diagnostic Perspectives. Biomolecules 2025;16(1);39
NEWLY APPROVED AND REGISTERED CLINICAL TRIALS AT THE CHR, - READY FOR ENROLLMENT
Please call our registration desk at (212) 994 4400 if you or a friend is interested in further information.