What is New Regarding the Polycystic Ovary Syndrome (PCOS)
By Norbert Gleicher, MD, Medical Director and Chief Scientist at the CHR. He can be reached through the editorial office of the Reproductive Times or, directly, at ngleicher(at)thechr.com
In today’s posting we present a broad overview about recent literature on the Polycystic Ovary Syndrome (PCOS). It seems like the subject is finally gaining some new and urgently needed traction and we hope to contribute by disseminating the new knowledge.
Please let us know what you think and – just as a reminder – we always welcome your comments and ideas and we – still – offer subscriptions to this publication for free.
The CHR’s Editorial Staff
MORE GENOMIC STUDIES ON PCOS SUBGROUPS/PHENOTYPES - That PCOS is likely not one single condition has been known for decades, even though agreement was never reached how many “phenotypes” the syndrome indeed represented. The most widely accepted classification (called Rotterdam criteria) defined PCOS by four phenotypes (A, B, C, and D), with especially some leading members of the Androgen Society having argued for the longest time that the D-phenotype should really be separated from the other three phenotypes.
More recently – using varying approaches and new research technologies the understanding of PCOS has shifted. Using clinical observations and genomic analyses, a new understanding of PCOS evolved: Over a decade ago PCOS – more or less accidentally - became a major research area at the CHR after we in preparation for a minor planned aging study of PCOS patients pulled all of the CHR’s PCOS patients out of the center’s electronic medical record data base and - to our surprise and really shock - discovered that the CHR almost exclusively had serviced only phenotype-D PCOS patients, - widely considered the smallest group of PCOS patients. Initially convinced that this must have been a coding error, we repeated the search, - only to confirm the results.
It then dawned on us that – as a last-resort fertility center (over 95% of CHR patients received and failed fertility treatments, usually IVF cycles, before reaching out to the CHR) – the A, B, and C – phenotype PCOS patients must be conceiving before reaching CHR, while something in the D-phenotypes must prevents these patients from conceiving even with IVF, which is why these women end up reaching the CHR in otherwise unheard proportions. Unsurprisingly, this conclusion activated our curiosity, and the rest is by now history.
Based exclusively on clinical observations in these patients the CHR’s investigators concluded that PCOS was basically a condition likely made up of only two phenotypes, a first representing mostly what under Rotterdam criteria was called phenotypes A, B, and C and – more-less in analogy of earlier suggestions by the Androgen Society – and a second group representing what had been the D-phenotype, also called the “lean” PCOS phenotype in contrast to all the others which are characterized by truncal obesity.
The CHR’s investigators – based only on clinical observations - were, therefore, the first to publish that PCOS likely represented only two distinctively different patient sub-groups with different relevance for fertility but also for their long-term well-being (there, of course, also exist overlapping cases).1,-3
In parallel the very prominent medical endocrinology group at Mount Sinai lead by Andrea E. Dunaif, MD, came to similar conclusions using genomic studies, with clustering studies also defining two PCOs phenotypes (i.e., clusters) and a third one representing a group that overlapped between those two, - forming a third cluster.4 They described these two groups as “reproductive” and “metabolic.” The former was characterized by high luteinizing hormone (LH), high sex hormone binding globulin (SHBG), and relatively low BMI as well as insulin levels, while the latter was characterized by high BMI, glucose and insulin levels and lower LH and SHBG.

In 2024 a group of Chinese investigators through genomic clustering also concluded that PCOS represented this kind of 2+1 distribution of phenotypes.5 Patients in this study underwent their first cycle of IVF and unsupervised cluster analysis was performed. Controlled ovarian stimulation parameters and reproductive outcomes were then compared between the different clusters of PCOS which they called “reproductive”, “metabolic”, and “balanced” clusters based on nine traits: The reproductive group was characterized by high levels of testosterone (T), SHBG, follicular stimulation hormone (FSH), LH, and anti-Müllerian hormone (AMH). The metabolic group was characterized by high BMI, fasting insulin, and fasting glucose. The balanced group was characterized by low levels of the aforementioned reproductive and metabolic parameters, except for SHBG.
Compared with reproductive and balanced clusters, those in metabolic cluster had lower rates of good quality day 3 embryo and blastocyst formation. Moreover, PCOS patients in the reproductive cluster had greater fresh embryo transfer (ET) cancelation rate and clinical pregnancy rate after fresh ET than metabolic cluster. And compared with PCOS of metabolic cluster, PCOS of balanced cluster also had higher chances for fresh ET cancelation.
And yet another most recent Chinese study in Nature Medicine suggested the presence of four “subtypes” which they validated across diverse populations 5 (as the CHR has done as well).5 Those four were: (i) Hyperandrogenism; (ii) Obesity; (iii) High SHBG; (iv) High LH as well as high AMH, - defined by unsupervised clustering of 9 clinical variables in 11,908 affected women (based on Rotterdam criteria) in a multicenter study validated across five international cohorts.
Despite some shortcomings (which we will return to below), this study offered unique insights because it included a 6.5 year follow up of patients and IVF treatments. Consequently distinct reproductive and metabolic trajectories could be observed. Those included the following: (i) Hyperandrogenic PCOS was associated with the highest prevalence of second trimester pregnancy loss and dyslipidemia; (ii) PCOS with obesity exhibited the most severe metabolic complications, lowest live birth rates and highest PCOS remission rates; (iii) PCOS with high SHBG demonstrated favorable reproductive outcomes and the lowest incidence of diabetes and hypertension; PCOS with high LH as well as AMH had the greatest risk of ovarian hyperstimulation syndrome (OHSS) and the lowest PCOS remission rates. Additional comments to the paper were offered in an accompanying Research Briefing by two of the authors in the same issue of Nature Medicine:7
A mild criticism considering an, otherwise, excellent stud is in place because by selecting their study population based on Rotterdam criteria, the researchers basically preempted the possibility of a “new” definition of PCOS. This study, therefore, just further defined the PCOS we all have known since establishment of the Rotterdam criteria in 2003. Characteristics of evolving patient subgroups are, however, quite amazingly similar between all the varying studies referred to here.
All here mentioned studies – despite obviously expected variability - have basically agreed on which clinical phenotypes represent characteristic presentations for the definition of PCOS subtypes/ phenotypes, - BMI defining obese vs, lean; androgen levels defining hyo- and hyperandrogenism; SHBG, physiologically going the opposite way of androgens; and beyond reproduction, defining long-term risk for metabolic syndrome (diabetes, hypertension, and heart disease, - Rotterdam phenotypes A, B, and C) and a hyperactive immune system as reported by the CHR (defined by autoimmunity, inflammation, and allergies, and Rotterdam phenotyp-D).
Most importantly for the infertility field, all of these publications moreover concur that some PCOS patients are affected in their reproductive success but not uniformly: The CHR believes that the clinical studies of the CHR defined these risks so-far the best when it comes to infertility, including IVF resistance, and with increased miscarriage risk by defining PCOS in two phenotypes (and an overlapping phenotype) along the following principles:
• All PCOS patients are characterized by abnormally high functional ovarian reserve (FOR) reflected by abnormally high AMH levels, antral follicle counts (AFCs) for age,and hyperandrogenism and, because of the latter’s effect on SHBG by normal or even low SHBG levels. This observation contradicts the Rotterdam criteria, which describe the D-phenotype as non-hyperandrogenic (an error we will explain next).
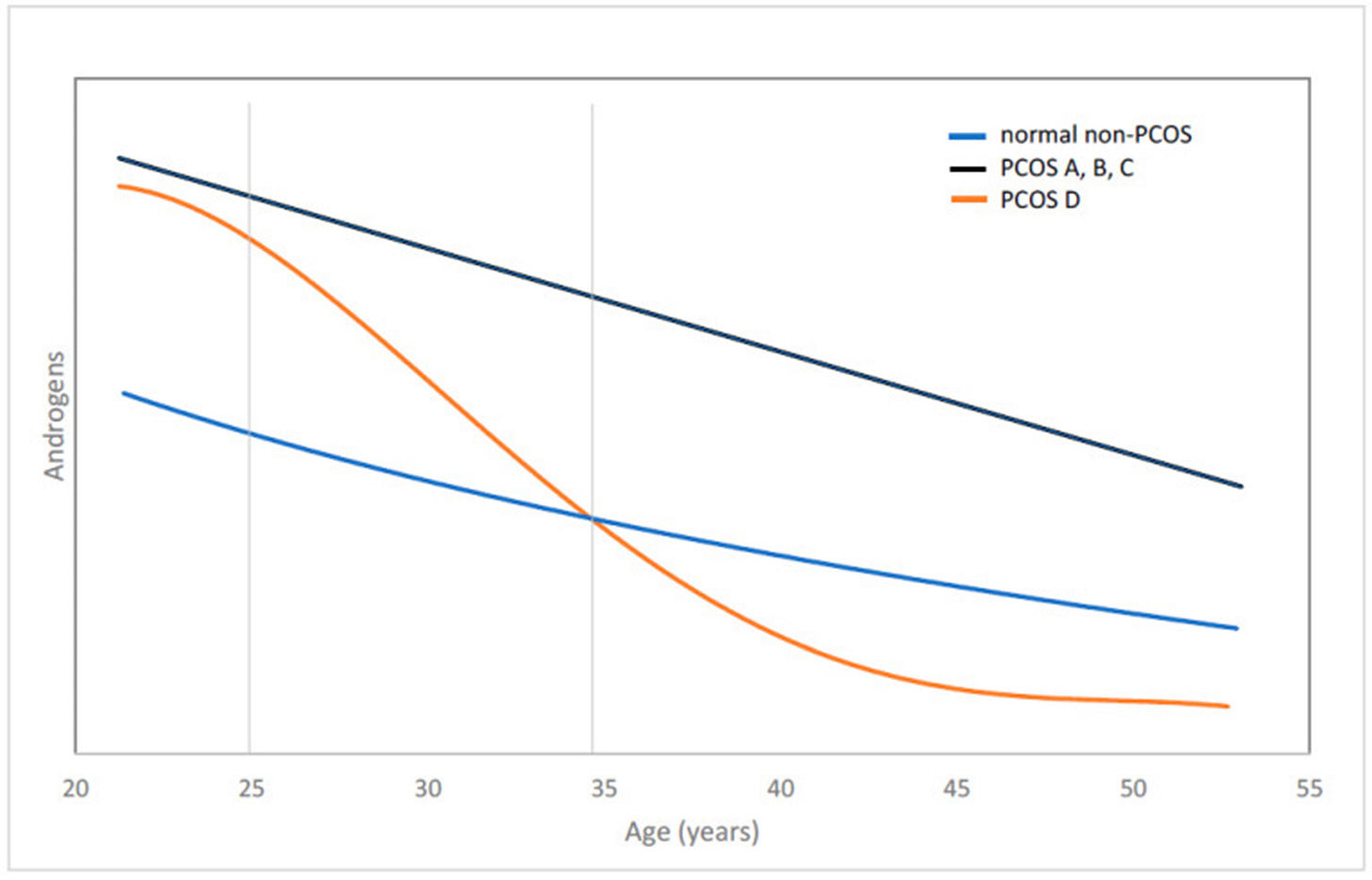
• The D-phenotype under Rotterdam criteria is hyperandrogenic, - but only short-term after menarche and – due to insufficiency of adrenal androgen production – starts a steady decline which, approximately by age 25 results in these women entering normal androgen range, where they remain for approximately a decade, only to reach hypo- androgenic levels by approximately age 35. Because over 90% of all PCOS diagnoses are made between ages 25 and 35, this is the reason why this phenotype by Rotterdam was described as normo-androgenic. All other PCOS patients remain hyperandrogenic into advanced ages.
• The D-phenotypes is correctly described by Rotterdam criteria as “lean” and usually ovulatory. These women, therefore, also are ovulatory (most of the other phenotypes are not) and therefore have regular menstrual patterns (other PCOS patients are usually obese and oligo-amenorrheic). The D-phenotype also does not present with hirsutism, acne and metabolic abnormalities (all stigmata routinely associated with PCOS. Consequently phenotype-D patients in a large majority of cases go undiagnosed or report a medical history of ones as teens having been told that they may be PCOS patients but later were told that this has been an erroneous diagnosis.
• This incorrect correction of their original diagnosis often moreover appears confirmed by the fact that these women – while in normal androgen range – are fertile and often normally conceive and deliver. They, however, often demonstrate a repeat miscarriage pattern, explained in a moment below.
• By approximately age 35 – after becoming hypo-androgenic – they suddenly become infertile. This is a major reason why women diagnosed as repeat (immune) aborters before that age demonstrate a natural history of becoming infertile. They, therefore, usually become infertility patients only after age 35 and, therefore, quickly enter IVF treatments, characterized by large egg numbers but, because of poor egg quality, few good quality embryos.
• This egg to embryo discrepance in numbers is a typical sign in the IVF history of these patients, is a consequence of their hypo-androgenism, and is the principal cause for their IVF resistance (there, of course, are also others). Because their hypo- androgenism is adrenal in nature (demonstrated by significantly lower DHEA-S than DHEA) and, therefore, correctable with androgen supplementation. Since hypo-androgenism is usually associated with elevated SHBG, successful androgen supplementation will reduce SHBG levels in parallel with increases in total testosterone (TT).
• Finally, PCOS is usually associated with risk for the metabolic syndrome. Women with D-phenotype usually do not experience this risk. They, however in ca. 85% of cases demonstrate a history and/or have laboratory evidence of a hyperactive immune system which can manifest as either autoimmune findings, inflammatory markers, and/or as significant allergies. They also in ca. 45% demonstrate evidence of autoimmune thyroid disease.
• Since hyperactive immune systems – whatever the cause – significantly predispose to immunological pregnancy loss, these women are at significantly increased miscarriage risk once they do conceive and must receive appropriate prophylactic treatments to prevent pregnancy loss. They also are at significantly increased risk for other pregnancy complications during a second high risk period in the third trimester of pregnancy, which mimic typical autoimmune risks, including premature labor and delivery, small for gestational age infants, preeclampsia and autoimmune exacerbation in the postpartum period.
The CHR has been publishing these facts now for over 10 years in medical journals1-3 and in our in-house publications, - the CHRVOICE and the Reproductive Times. We, therefore, are still always surprised how many undiagnosed patients with this presentation still present to the CHR as a practically daily event. Because these women go from being hyper- to being hypo-androgenic as a key diagnostic feature (the other one being laboratory evidence of a hyperactive immune system), we have described this PCOS phenotype/ subgroup as hyper/hype-PCOS (HH-PCOS).
Here cited publications of genomic studies by the Mount Sinai group in NYC – just a few blocks uptown from the CHR on Madison Avenue and, since then, from two Chinese groups – while not 100% overlapping with our clinical observations – are nevertheless strongly supportive of the CHR’s understanding of how PCOS affect fertility and reproductive success in general and represents one of the most important breakthroughs the CHR achieved in its over 40 years-long research history, marked by several major breakthroughs.
It is also an excellent example for two additional observations worthwhile mentioning: First, even in days of amazing genomic capabilities, clinical observations are, still, at the core of medical practice. And one can never predict where an unexpected finding will lead to. In this case, indeed, two totally unexpected findings lead to the discoveries here described: The unexpected discovery that practically all of the CHR’s PCOS patients were phenotype D patients, we already mentioned. A second one – not less important, - however, also deserves recognition: Preceding everything we here presented, the CHR only once in its decade-long history submitted a paper to a prestigious medical journal in which its investigators could not explain the outcome of the reported study.
And we said so in the paper, even though we were concerned that the journal for that reason would not accept the paper for publication. The editor-in-chief of the journal at the time was, however, pleasantly surprised us and (wisely!) accepted the paper.8
That paper reported on the CHR’s IVF cycle outcomes based on how FSH and AMH levels in their relationship affected pregnancy chances. Both of these hormones are supposedly assessing FOR. They, therefore, should run in parallel, - that is, of course, in opposite directions - as FSH should go up and AMH should go down as FOR declines. But we observed was that this not always was the case. To our surprise, we found that a HIGH/HIGH pattern gave us by far the best pregnancy rates and we had no idea why?
That is, - until we started to get interested in above described PCOS research. It suddenly dawned on us why the HIGH/HIGH pattern had done so well: Those women were practically all women with PCOS (high AMH). And they were HH-PCOS patients because we did not see any other PCOS patients at the CHR.
By that time (2012), we had already for a good number of years been supplementing hypo-androgenic women with DHEA, which very frequently improves egg quality and pregnancy chances because ovaries need good androgen levels to make good eggs (another IVF break through made at the CHR9). And since we now understand what our 2013 paper in the Journal of Clinical Endocrinology and Metabolism (JCEM) really meant, we now had another diagnostic hint when a new patient presented with elevate FSH (which happens quite often in PCOS patients) but also has higher AMH than one would expect with an elevated FSH.
And this also needs to be said: The CHR is incredibly lucky for having the most unique patient population of any IVF clinic in the U.S., and likely in the world. If it were not for this unique quality of the CHR’s patients, our investigators could never have made all of the breakthroughs in fertility treatments the CHR has achieved and is continuing to make. A big Thank You, therefore, goes to all of our past and future patients and to our physicians and other scientists for always going with open eyes through the day.
ANOTHER PAPER WITH CONSIDERABLE RELEVANCE TO THE PRECEDING – And since we have been already talking about PCOS phenotypes, the Mount Sinai Endocrinology group, and the JCEM, here is some information about another article in JCEM by the Mount Sinai group in which they report the ontogeny of distinct reproductive phenotypes in girls at risk for PCOS during the postmenarchal transition.10
Daughters of women with PCOS and girls with overweight/obesity can have hyperandrogenemia beginning in childhood. Other features of their early reproductive phenotypes, however, can differ, suggesting the existence of mechanisms conferring upon them increased risk for PCOC. Unfortunately investigating only a too small a number of children (PCOS 15, overweight 12, and 17 lean controls) between 0.2 to 1.2 years of menarche the number of patients was really too small to be able to come to definite conclusions. The data, however, were, nevertheless, interesting.
As one would expect, SHBG levels were lower in PCOS and overweight girls than in lean controls. Free testosterone levels were higher in overweight girls, while DHEA-S levels were marginally higher in PCOS vs. controls and trended higher in overweight girls. Morning LH levels were higher in PCOS vs overweight girls and LH and FSH responses to GnRH analog were also increased in PCOS vs overweight girls. The prevalence of hyperandrogenemia was similarly increased with PCOS and overweight, - and the prevalence of ovulatory dysfunction was also increased in both groups.
The authors concluded that PCOS and overweight girls had persistent hyperandrogenemia during the early postmenarchal transition. However, ovulatory dysfunction, neuroendocrine abnormalities, and elevated basal and stimulated LH responses to GnRH analog were observed only with PCOS and suggested that these findings support the existence of distinct developmental trajectories leading to polycystic ovary syndrome, with early neuroendocrine dysregulation in PCOS and with overweight to peripheral, likely adiposity-related, androgen excess .
This was neither a very exiting paper in content nor in recognition of preceding publications. For example, a 2016 paper by NIH investigators from what then was the Stratakis laboratory, reported on a subset of young women with PCOS with a pattern of glucocorticoid secretion that mimicked that of patients with micronodular adrenocortical hyperplasia.11
This meant that they had smaller adrenal volumes and higher steroid hormone secretion after dexamethasone compared to a group of PCOS patients with appropriate response to dexamethasone. This possibly also meant that micronodular adrenocortical hyperplasia could be a precursor condition to above -discussed PCOS phenotype D.
AND A MINI-REVIEW ON THE INTIMATE RELATIONSHIP OF ADIPOSITY TO PCOS BY TWO GIANTS IN PCOS RESEARCH – Worldwide, there are very few other people around with the credibility of Robert L. Rosenfield, MD, and Daniel A. Dumesic, MD., the first a pediatric endocrinologist in San Francisco and the second a REI in Los Angeles. Any Review article for which these two powerhouses get together, therefore, is worth the read.
And the recent Mini Review in the JCEM on adiposity in association with PCOS is certainly no exception.12 It is, however, important to point out that the authors – wisely – restricted their review to “the relationship of increased adiposity to hyperandrogenic oligo-anovulatory PCOS.”
Why is this important? Because most papers in the literature, unfortunately, do not distinguish between PCOS phenotypes in selecting patient populations for studies or in discussing PCOS. By concentrating their review only on hyperandrogenic PCOS, they for all practical purposes did not include above extensively discussed D-phenotype under Rotterdam criteria (even though – as also noted above – this phenotype for a short period between menarche and ca. age 25 is also hyperandrogenic) and this kind of distinction between PCOS subgroups/phenotypes is, of course, what must finally happen, - if highly overdue progress is to be made in better understanding PCOS.
The authors, indeed, implicitly endorse the so-called two “reproductive” and “metabolic” subtypes of PCOS as described by the Mount Sinai group4 when noting that PCOS demonstrates functionally two forms, a “functional typical” form characterized by ovarian hyperandrogenism characterized by a unique pattern of ovarian steroidogenic hyperresponsiveness to gonadotropin stimulation explainable by gene variants that cause overexpression of an activating variant of DENND1A and of a “functional atypical” form (roughly one-third of PCOS cases) lacking this excessive ovarian response.
While the authors claim that their “typical” and “atypical” forms of PCOS share clinical traits with the respective “reproductive” and “metabolic” subtypes described by the Mount Sinai group, we would argue that this alleged overlap is much less pronounced than the overlap in clinical features between the two Sinai group phenotypes and the two by the CHR reported phenotypes. We would further argue that the difference in overlaps in those two comparisons is, likely due to inclusion of patients in studies which, individually, showed overlaps in clinical presentation between the two principal sub-groups/phenotypes, thereby establishing, for practical purposes a third “mixed” group.
Returning to the main subject of the Mini Review (we reemphasize here that the authors specifically only addressed hyperandrogenic PCOS), the authors suggested preferential, often subclinical abdominal fat accumulation represents a central feature of PCOS that also affects metabolic functions.
Here is, however, an important additional point that this paper – and really practically all PCOS research – still is overlooking: For infertility practice (we are here not addressing long-term health effects of PCOS) the “classical” hyperandrogenic, obese, anovulatory PCOS patient is – as the CHR’s patient experience so well demonstrates - a solved problem. These women conceive with standard fertility treatments, whether ovulation induction alone or IVF. The remaining problem are the PCOS patients who do not conceive, and that is the HH-PCOS patient!
A WORD ON THE IMMUNE SYSTEM IN PCOS – That PCOS can be associated with immune system abnormalities has been known for decades. A detailed understanding has, however, over all this time been prevented because PCOS was never investigated sub-group/phenotype -specific. To the best of our knowledge, this was for the first time done when the CHR’s investigators noted the approximately 85% association of the HH-PCOS phenotype with a hyperactive immune system in general and with thyroid autoimmunity in ca. 45%.1-3
Now a group of investigators from the U.S. closely associated with the Androgen Society (though in this paper not speaking for the society) offered some additional information in a paper in JCEM.13 Importantly, this retrospective cross-sectional study used blood samples from the Androgen Excess Biorepository, according to the British Society for Endocrinology a specialized collection of biological samples such as blood, serum, and tissues, —paired with clinical data, dedicated to researching conditions where androgenic steroids are elevated, primarily PCOS. These repositories, therefor, primarily are geared toward the study of hyperandrogenism which, of course, automatically biases every PCOS study since it almost completely excludes normo- and hypo-androgenic PCOS patients (i.e., the HH-PCOS phenotype above age 35). Because the study population involved women between ages 18 and 45 years, between ages 25 and 35, a small group of still hyper-androgenic young women may have been included. However, their long-term disease profile would, however, have separated them from prolonged hyperandrogenic PCOS patients.
Because these two groups of patients were not separately analyzed, in this paper presented data must be viewed with caution. It is, indeed, reasonable to assume that – given what the CHR reported the high prevalence of hyperactive immune systems to be in HH-PCOS patients, the in the study obtained results would probably have looked differently, - had HH-PCOS patients been removed from consideration before analysis or had been analyzed separately.
The study results, however, suggested the following: What the authors described as a significantly lower level of circulating immune markers (CRP, 96 markers including IL-6, TNF-alpha, IL-18, interferon-gamma) was really only a marginal result (p<0.05). Growth factors, proinflammatory cytokines, and chemokines were – independent of race – reduced. The authors concluded from these findings that what they considered to be a prevailing notion to be, - namely that PCOS is a chronic low-grade inflammation -was incorrect. Instead, they are now considering PCOS a condition of immune suppression and impaired angiogenesis.
Studying this paper, one, indeed, does not know where to begin the criticism because everything – from patient selection and description to data interpretation and conclusions – is (trying to remain polite) insufficient. This paper therefore is, indeed, a very good example for why PCOS has remained such a confusing diagnostic entity and why reproductive immunology has gotten such a bad reputation in reproductive medicine.
MYOINOSITOL (MI) IN THE CLINICAL MANAGEMENT OF PCOS – A recent Commentary by Italian colleagues in RBM Online confirmed our suspicion that MI has slowly been gaining widespread use in the management of infertility in PCOS patients.14 And once again, one has to wonder why and based on what studies?
As the authors state in their article, there exist claims that inositols improve insulin sensitivity and ovarian function. But where is the evidence?
The authors, therefore, correctly conclude that myoinositol (and for that matter also other inositols) should not be recommended as routine treatment in PCOS patients. We, of course, fully concur but want to make an additional point we have repeatedly made in the past in these pages: Myoinositol should definitely not be used in normo- and hypo-androgenic PCOS patients because it lowers androgen levels even further.
VITAMIN D IN IVF CYCLES OF WOMEN WITH PCOS – Yet another Chinese multicenter study – this time in the BMJ – which investigated whether vitamin D supplementation would improve IVF outcomes in PCOS patients.15 And to be blunt, not only did vitamin D supplementation not show any effects on IVF cycle outcomes in PCOS patients, - but the paper really did not even offer a hypothesis why vitamin D should be effective.
Moreover, the study design can only be described as pathetic (yes, even though the paper appeared in the BMJ, at least by some in the medical field still consider a respectable medical journal): (i) PCOS phenotypes, of course, were not separated. They, indeed, were not even noted. (ii) Out of 1,272 patients screened, only 31 were found to be ineligible but 365 declined participation (one wonders why and how much their refusal further biased the study). (iii) That left 876 alleged PCOS patients (per Rotterdam Criteria and, therefore, including phenotype-D patients, - i.e., HH-PCOS patients, - though in untold distribution) which means 36.5 patients only per participating center (24 clinics participated). Imagine how different IVF cycle management protocols must have been between these centers.
In other words, just one more of these completely useless studies that appear to continue to increase in numbers and are giving medical and scientific research a bad name at a time, when especially some Chinese research laboratories have really started to produce worldclass work.
REFERENCES
Kushnir et al., J Ov Res 2015;8:45. Doi. 10.1186/s13048-015-0175-x.
Gleicher et al., Endocrine 2018;59(3):661-676
Gleicher et al., Biomedicines 2022;10(7);1505
Dapas et al., PLoS Med 2020;17.e1003132
Chen et al., Chin Med J. 2024;137(5):604-612
Gao et al., Nat Med 2025;31:4214-4224
Zhao S, Zhao H. Nat Med 2025;31:4002
Gleicher et al., J Clin Endocrinol Metab 2013;2136-2145
Gleicher N, Barad DH. Hum Reprod 2008;23(12):2868-2870
Torchen et al., J Clin Endocrinol Metab 2026;111:553-560
Gourgari et al., J Clin Endocrinol 2016; 101(9):3353-3360
Rosenfield RL, Dumesic DA. J Clin Endocrinol Metab 111(1):11-23
Pacheco-Sanchez et al., J Clin Endocrinol Metab 2026;111:656-664.
Palomba et al., RBMOnline 2026;52(2):105269
Hu et al., BMJ 2026392:e087438.
NEWLY APPROVED AND REGISTERED CLINICAL TRIALS AT THE CHR, - READY FOR ENROLLMENT
Please call our registration desk at (212) 994 4400 if you or a friend is interested in further information.